Elective atrial septostomy (AS) can provide additional clinical benefits to adults receiving targeted triple therapy (endothelin receptor antagonists, phosphodiesterase-5 inhibitors, and prostanoids) for severe pulmonary arterial hypertension (PAH). Elective AS can be used as a bridge to lung transplantation when medical treatment is ineffective or as a definitive treatment when transplantation is contraindicated. Clinical guidelines recommend AS for PAH patients in functional class III or IV with refractory right heart failure or severe exercise-induced syncopal symptoms, either as a pretransplant treatment or as a palliative measure when no other treatment options are available.1 However, this intervention has been little used in our setting.
We present experience with elective AS in PAH patients under targeted triple therapy at a referral center. To our knowledge, this is the largest series in Spain describing results obtained with this technique in the context of current PAH treatment.
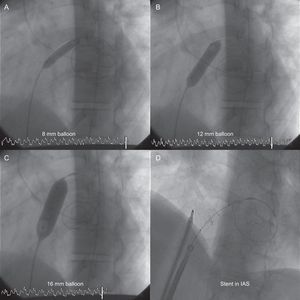
Since 2005 we have performed 11 balloon AS procedures and 1 stent fenestration of the atrial septum in a total of 10 patients (8 women, mean age 43.5 years). Patient data and procedures are summarized in the Table. All patients were in PAH functional class III or IV and all were taking epoprostenol, either in targeted triple therapy with endothelin receptor antagonists and phosphodiesterase-5 inhibitors (7 patients), or in double therapy with phosphodiesterase-5 inhibitors (3 patients). Patients were prepared for AS by blood transfusion and treatment with inotropes and intravenous diuretics. Elective AS was not used as a rescue procedure in end-stage disease. A transseptal puncture was made with a Brockenbrough needle followed by gradual balloon dilatation, according to the protocol described by Sandoval et al.2 The procedure was carried out without sedation in order to properly monitor changes in peripheral artery oxygen saturation. As stipulated in the protocol,2 the transseptal opening was expanded gradually, initially with a balloon diameter of 4mm and then sequentially with balloons dilated to 8, 12, and 16mm. Each dilatation was followed by a 3minute pause, at the end of which readings were taken of left ventricular end-diastolic pressure and arterial oxygen saturation. Final aperture size was determined individually for each patient, with dilatation stopped when any of the following conditions were met: elevation of left ventricular end-diastolic pressure > 18mmHg, reduction in peripheral artery oxygen saturation to < 80% or by > 10% relative to the basal level, or dilatation of the interatrial septum to the maximum aperture (16mm). The procedure was guided by intracardiac echocardiography in 2 patients and by fluoroscopy alone in the others. For the patient who underwent stent fenestration of the interatrial septum, we used the diabolo-shaped stent configuration described by Stümper et al,3 without sedation and with monitoring by fluoroscopy and intracardiac echocardiography. After making the transseptal puncture with a Brockenbrough needle, an Inoue guide wire was advanced into the left atrium and the interatrial opening was dilated with a dilator and a Mullins sheath. This was then replaced with a 12 French sheath, while maintaining the Inoue guide wire in the left atrium. A 19mm Palmaz® GenesisTM stent (Johnson & Johnson) was mounted on a 16mm × 40mm valvoplasty balloon, around which a central 5mm diameter bow was tied with an epicardial pacing wire. The mounted stent was delivered through the sheath along the Inoue guide wire. Fluoroscopy and intracardiac echocardiography guidance was used to center the pacing wire loop at the opening of the interatrial septum, and the balloon was inflated. The pacing wire loop restricts expansion of the center of the stent and thus results in a diabolo-shaped configuration centered across the atrial septum; the fenestration diameter is predetermined by the diameter of the pacing wire loop around the balloon. In all patients, balloon AS resolved refractory right heart failure and eliminated exercise-induced syncope. The first patient developed complete atrioventricular blockade. No other complications were encountered with the technique. The mean follow-up period was 24 months (range, 1-67 months). During follow-up, the transseptal fenestration closed spontaneously in 3 patients (33%): in 1 patient the peripheral artery oxygen saturation contraindicated repeat AS, in another we performed stent fenestration of the atrial septum (Figure), and in the third we performed a repeat AS at 18 months. Overall survival was 90% at 30 days, 75% at 6 months, and 57% at 1 year. One patient died 1 week after AS during anesthesia for implantation of a Hickman catheter; 3 others died due to disease progression. Of the surviving patients, 3 underwent successful transplantation, 2 were on the transplant waiting list, and the patient with the stent fenestration of the interatrial septum showed no reclosure or device migration after 44 months.
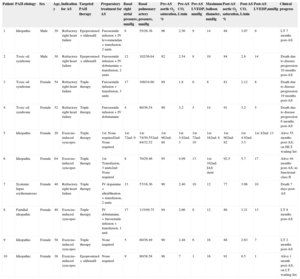
Patient Data and Atrial Septostomy Procedures
| Patient | PAH etiology | Sex | Age, y | Indication for AS | Targeted PAH therapy | Preparatory treatment for AS | Basal right atrial pressure, mmHg | Basal pulmonary artery pressure, mmHg | Pre-AS aortic O2 saturation, % | Pre-AS CO, L/min | Pre-AS LVEDP, mmHg | Maximum balloon diameter, mmHg | Post-AS aortic O2 saturation, % | Post-AS CO, L/min | Post-AS LVEDP,mmHg | Clinical progress |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Idiopathic | Male | 39 | Refractory right heart failure | Epoprostenol + sildenafil | Furosemide infusion + IV levosimendan + transfusion, 2 units | 8 | 55/26.36 | 96 | 2.56 | 9 | 14 | 88 | 3.07 | 9 | LT 7 months post-AS |
| 2 | Toxic oil syndrome | Male | 30 | Refractory right heart failure | Epoprostenol + sildenafil | Furosemide infusion + IV dobutamine + transfusion, 2 units | 12 | 102/36.64 | 92 | 2.54 | 8 | 10 | 84 | 2.8 | 14 | Death due to disease progression 10 months post-AS |
| 3 | Toxic oil syndrome | Female | 54 | Refractory right heart failure | Triple therapy | Furosemide infusion + transfusion, 3 units | 17 | 100/34.60 | 89 | 1.8 | 6 | 8 | 81 | 2.12 | 8 | Death due to disease progression 19 months post-AS |
| 4 | Toxic oil syndrome | Female | 52 | Refractory right heart failure | Triple therapy | Furosemide infusion + IV dobutamine | 4 | 80/36.54 | 90 | 3.2 | 5 | 14 | 91 | 3.2 | 5 | Death due to disease progression 6 months post-AS |
| 5 | Idiopathic | Female | 20 | Exercise-induced syncopes | Triple therapy | 1st: None required2nd: None required | 1st: 72nd: 9 | 1st: 74/30.552nd: 84/32.52 | 1st: 962nd: 88 | 1st: 3.92nd: 3 | 1st: 72nd: 10 | 1st: 162nd: 8 | 1st: 902nd: 82 | 1st: 4.92nd: 3.5 | 1st: 82nd: 13 | Alive 55 months post-AS; on HLT waiting list |
| 6 | Idiopathic | Female | 64 | Exercise-induced syncopes | Triple therapy | 1st: Transfusion, 3 units2nd: None required | 8 | 70/29.46 | 95 | 4.99 | 13 | 1st: 162nd: IAS stent | 92.5 | 5.7 | 17 | Alive 44 months post-AS; in functional class II |
| 7 | Systemic lupus erythematosus | Female | 40 | Refractory right heart failure | Triple therapy | IV dopamine + ultrafiltration + transfusion, 2 units | 13 | 57/16.36 | 90 | 2.40 | 10 | 12 | 77 | 3.96 | 10 | Death 7 days post-AS |
| 8 | Familial idiopathic | Female | 40 | Exercise-induced syncopes | Triple therapy | IV dobutamine. + furosemide infusion + transfusion, 1 unit | 17 | 115/49.75 | 94 | 2.66 | 8 | 12 | 86 | 3.21 | 13 | LT 8 months post-AS |
| 9 | Idiopathic | Female | 58 | Exercise-induced syncopes | Triple therapy | None required | 5 | 80/36.49 | 90 | 2.48 | 6 | 16 | 88 | 2.63 | 7 | LT 2 months post-AS |
| 10 | Idiopathic | Female | 38 | Exercise-induced syncopes | Epoprostenol + sildenafil | None required | 9 | 80/38.58 | 98 | 7 | 1 | 16 | 91 | 8.5 | 1 | Alive 1 month post-AS; on LT waiting list |
AS, atrial septostomy; CO, cardiac output; HLT, heart and lung transplant; IAS, interatrial septum; IV, intravenous; LT, lung transplant; LVEDP, left ventricular end-diastolic pressure; PAH, pulmonary arterial hypertension.
The utility of AS in patients receiving targeted triple therapy for PAH has not been evaluated. In most published series, only a very low percentage of patients received targeted triple therapy.4,5 In our series, all patients received epoprostenol, 7 of them in targeted triple therapy and 3 in double therapy with phosphodiesterase-5 inhibitors. Even in the context of this optimal treatment, AS provided additional clinical benefit in all patients, improving functional class and resolving refractory right heart failure and syncopes. These benefits were achieved both when AS was used as a preparation for lung transplantation and in patients in whom medical treatment had failed and there were no therapeutic alternatives. The failure of AS to become established as a general treatment is likely due to the relatively high associated procedural and periprocedural mortality (5% and 16%, respectively),5 and also to uncertainty about the optimal timing of the intervention for maximum benefit. Some studies suggest that AS would be beneficial at early stages of disease development; nonetheless, in most countries with access to high-cost medical treatments and lung transplantation, AS is conducted at late disease stages, when medical treatment has failed or as a last resort before transplantation. However, AS in terminal disease stages is associated with high mortality, and the procedure is therefore not recommended in end-stage patients.4,5 In our series, AS was indicated in advanced disease stages when medical treatment had failed, but not as a rescue treatment in end-stage patients. The patient who died 1 week after AS had needed ultrafiltration and inotrope therapy as preparation for the procedure, and it is possible that the indication for AS was borderline in this case. No patient died as a result of the procedure. Survival in our series at 30 days was 90%, and at medium term was in line with published data related to disease progression: 75% at 6 months and 57% at 1 year.1,4,5
In our experience, elective AS is a therapeutic option for PAH patients in whom optimal medical treatment has failed. The procedure is associated with low periprocedural morbimortality, and delivers functional improvement in most severe PAH patients with refractory right heart failure or low cardiac output. Moreover, elective AS is effective both as a bridge to lung transplantation and as an adjunct to medical treatment.