Electrical storm (ES) is defined as the appearance of 3 or more episodes of ventricular tachycardia or fibrillation within 24hours requiring antitachycardia therapy or cardioversion/defibrillation.1 In practice, the number of episodes is usually much higher, producing an extremely serious situation. The role of the sympathetic nervous system in ES is well established,2 and sympathetic blockade has been shown to effectively control these situations.3 The first studies with sympathetic blockade centered on its use to treat congenital long-QT syndrome in patients unresponsive to beta-blocker therapy, and it has recently been applied in the treatment of ES.4
The goal of this study was to present our accumulated experience with transient sympathetic blockade (TSB) in the treatment of a series of patients with refractory ES, defined as ES persisting after conventional therapy for the control of ES and its immediate causes.
Refractory ES was diagnosed according to the judgment of the responsible physician, without prior specification of criteria for episode duration, number, or toleration. In all patients, TSB was first attempted by left stellate ganglion block (LSGB); patients with recurrent ES or poor initial control after LSGB were scheduled for bilateral block by thoracic epidural anesthesia. LSGB was performed by ultrasound-guided bolus injection of local anesthetic via the paratracheal route. The procedure was carried out at the patient's bedside in the coronary care unit by anesthesia unit staff. Ultrasound guidance was also used to place a soft catheter for continuous infusion of 0.2% ropivacaine. The mean infusion rate was 7mL/h, with an initial rate of 6mL/h and a maximum of 12mL/h. The efficacy of TSB was evaluated by clinical observation: the efficacy indicators were clinical signs of Horner syndrome for LSGB and relief of anterior chest pain in patients given thoracic epidural anesthesia. In all patients, TSB was used as an addition to conventional antiarrhythmic therapy and in no instances replaced or required withdrawal of baseline pharmacologic treatment. In 2 patients, LSGB did not achieve sufficient electrical control, and these patients were given thoracic epidural anesthesia with 0.2% ropivacaine. Sympathetic blockade was withdrawn according to the medical team's criteria after the patient had been free of arrhythmias for 48hours.
The effectiveness of the technique was estimated by comparing the number of episodes of sustained ventricular arrhythmia before and after TSB. The statistical significance of the reduction in the number of episodes after LSGB was assessed by the Wilcoxon test for paired data.
Between March 2012 and December 2014, our team performed TSB on 8 patients diagnosed with refractory ES. The mean patient age was 58 years, and 75% had severe left-ventricular systolic dysfunction. Baseline patient characteristics, the trigger for ES, and the initial treatment are summarized in the Table.
Baseline Patient Characteristics, Electrical Storm Trigger, Initial Treatment, and Follow-up
| Age, y | Sex | Underlying disease | Trigger | Type of arrhythmia | ICD | LVEF, % | TI | IABP | Drugs | Technique for transient SB | Duration of SB, d | Coronary angiography | Revascularization | In-hospital death | Cause of death | Recurrence of ES | Follow-up, time | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 57 | Male | Ischemic heart disease | Heart failure | Polymorphic VT | No | 12 | Yes | Yes | BB, amiodarone, lidocaine | LSGB | 5 | No | No | No | No | Alive, no readmissions (33 months) | |
| 2 | 81 | Male | Valvular heart disease | Acquired long QT syndrome | Polymorphic VT | No | 60 | Yes | No | Magnesium | LSGB | 4 | No | No | Yes | Sepsis - VAP | No | Deceased |
| 3 | 58 | Male | Dilated cardiomyopathy | Sepsis/heart failure | Polymorphic VT | Yes | 26 | No | No | BB, amiodarone | LSGB | 6 | No | No | Yes | Heart failure | No | Deceased |
| 4 | 58 | Male | Dilated cardiomyopathy | Ventilator withdrawal | Polymorphic VT | No | 31 | Yes | No | BB, amiodarone, procainamide | LSGB | 10 | Yes | No | No | Yes (7 days) | Alive, no readmissions (13 months) | |
| 5 | 58 | Male | Dilated cardiomyopathy | Heart failure | Polymorphic VT | Yes | 25 | Yes | No | BB, procainamide | LSGB/TEA | 9/6 | No | No | No | No | Alive, heart transplant (12 months) | |
| 6 | 71 | Male | Ischemic heart disease | Heart failure | Polymorphic VT | Yes | 15 | Yes | No | Amiodarone | LSGB | 6 | Yes | No | Yes | Ventricular arrhythmia | Yes (3 days) | Deceased |
| 7 | 49 | Female | No structural heart disease | Unknown | Polymorphic VT | No | 61 | No | No | BB, amiodarone | LSGB/TEA | 2/2 | Yes | No | No | No | Alive, no readmissions (2 months) | |
| 8 | 58 | Male | Ischemic heart disease | Heart failure | Polymorphic VT | No | 25 | Yes | Yes | BB, amiodarone | LSGB | 9 | Yes | No | Yes | Arterial ischemia | Yes (11 days) | Deceased |
BB: beta-blockers; IABP: intra-aortic balloon pump; ICD: implantable cardioverter-defibrillator; LSGB: left stellate ganglion block; LVEF: left-ventricular ejection fraction; SB: sympathetic blockade; TEA: thoracic epidural anesthesia; TI: tracheal intubation; VAP: ventilator-associated pneumonia; VT: ventricular tachycardia.
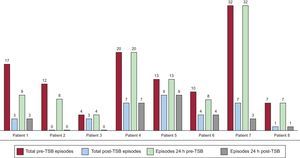
In 6 patients, the number of arrhythmia episodes declined during the first 24hours after the initiation of sympathetic blockade (Figure). In all 6 patients, LSGB significantly reduced the total number of episodes and the number of episodes in the first 24hours after the intervention (P < .05). Electrical storm recurred in 3 patients (37%) during the first 24hours after the cessation of sympathetic blockade. Of these patients, 2 responded to repeat LSGB. The third patient responded to conventional therapy and thus there was no further requirement for sympathetic blockade.
Mortality was 50% (patients 2, 3, 6, and 8), but only 1 death (patient 6) was due to arrhythmia. Catheter ablation of ventricular tachycardia was performed in patient 4, and patient 5 underwent heart transplantation. The follow-up of each patient is presented in the Table. In all patients, sympathetic blockade was transitory and its use did not interfere with conventional treatments, including definitive treatments such as catheter ablation and heart transplantation. There were no complications related to the procedure.
The results of our study suggest that TSB is a useful technique for the control of refractory ES. In our study, we used standard techniques for transient sympathetic blockade without having to resort to definitive methods. This contrasts with other studies reporting the use of interventions such as surgical sympathectomy5 or videothoracoscopy ablation of the sympathetic chain ganglia in the pleural cavity.
The main limitations of our study are the small sample size and its observational and retrospective nature. In addition, due to patient care and organizational issues, it was not possible to retrospectively determine the exact number of patients presenting with refractory ES during the study period (March 2012 to December 2014).