Keywords
INTRODUCTION
Left ventricular systolic dysfunction as a cause of arrhythmia is a well-recognized clinical entity that was initially reported in association with incessant supraventricular tachycardia in children and adolescents,1 and later with atrial fibrillation.2 Less commonly known is the fact that ventricular arrhythmia can induce ventricular dysfunction.3-11 In this article we describe 5 clinical cases of repetitive monomorphic ventricular arrhythmia in patients with no structural heart disease, except for left ventricular systolic dysfunction. Ventricular function improved considerably after radiofrequency ablation was performed.
PATIENTS AND METHODS
Between November 1997 and November 2003 in our unit, 37 patients with ventricular tachycardia and no known underlying pathology were studied. Five of these patients, whose main characteristics are shown in the Table, had left ventricular systolic dysfunction. The clinical diagnosis was dilated cardiomyopathy in 4 cases and myocarditis in 1. The ventricular arrhythmia consisted of high-density ventricular ectopic activity associated with runs of repetitive nonsustained ventricular tachycardia in 4 cases (Figure 1). Ablation was indicated on the basis of palpitations in four patients, and was further supported in one of them (Case 1) by the fact that the patient's ventricular function showed evidence of deterioration 11 months after the arrhythmia was detected. In Case 4 there were no palpitations and ablation was performed for suspected tachycardia-induced cardiomyopathy, based on our previous experience. In all cases coronary disease was excluded by angiography.
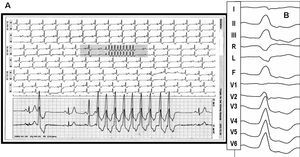
Figure 1. Panoramic view of electrocardiographic monitoring of Patient 2 in the left panel. Constant ventricular bigeminy and a run of ventricular tachycardia are evident. The enlarged tachycardia tracing (A) shows that the complexes have the same morphology as the bigeminal beats. The right panel (B) shows the morphology of the premature beats in the 12-lead ECG.
After the diagnostic electrophysiological study, activation mapping was performed starting with the right ventricle in all patients except Case 5. In this patient the initial approach in the left ventricle suggested by the electrocardiographic pattern (RS in V2), was ineffective; hence an irrigated catheter was successfully inserted in the proximity of the tricuspid ring.
After ablation, M-mode echocardiography was used to measure the dimensions of the left ventricle, and the ejection fraction was calculated with the Teichholz formula. Follow-up echocardiography was performed 7±2 months later. Long-term follow-up (4-69 months) included Holter monitoring in addition to the usual examinations.
RESULTS
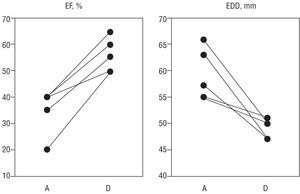
Ablation with a mean of 5.4 radiofrequency applications (range, 2-10) was performed without complications (Figure 2). No recurrence of the primitive ventricular arrhythmia was detected during follow-up. In 2 cases isolated ventricular premature beats with a different morphology were detected. All patients were asymptomatic at the most recent follow-up visit and were not receiving medication, except for 2 patients who were taking antihypertensive drugs. Post-ablation echocardiography showed an evident increase in the ejection fraction and a decrease in the left-ventricular end-systolic dimension in all 5 cases (Figure 3).
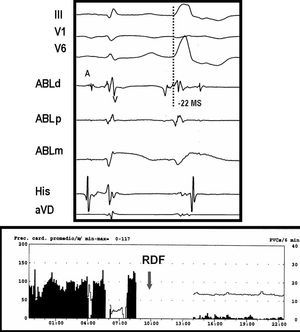
Figure 2. Tracing of the same patient as in Figure 1. The upper panel shows the intra-atrial electrograms of a premature ventricular beat recorded at the point of successful ablation in the left ventricular outflow tract. In the bipolar electrode (ABLd), early activation (22 ms) with respect to the QRS pattern of the surface leads is apparent; in the monopolar electrode (ABLm) a QS pattern can be seen. The lower panel shows suppression of the premature beats after radiofrequency ablation.
Figure 3. The left ventricular ejection fraction (EF) increased dramatically in all patients after suppressing the arrhythmia, and the end-diastolic dimension (EDD) decreased, indicating favorable ventricular remodeling. Baseline data (left in both panels) were obtained within the first 48 hours post-ablation. Data on the right were obtained several months after.
DISCUSSION
The main interest of the cases presented resides in the fact that ventricular dysfunction induced by ventricular arrhythmia substantially improved with abolition of the arrhythmogenic source. All the previous publications on this subject are individual case reports, with the exception of the study by Grimm et al,10 which included four patients. The first of the case reports was published in 1989 and the treatment consisted of antitachycardia pacemaker placement.3 In the remaining cases, radiofrequency ablation resolved the repetitive tachcardia4,5,7,10 or the frequent ventricular premature beats8,11 originating in the right ventricular outflow tract, except in 2 cases.6,9 The patients in our series had nonsustained monomorphic arrhythmia, but with a repetitive, incessant character. The location of the source varied, but did not impede successful ablation in all 5 cases.
It is more difficult to establish suspicion of this entity than when dealing with tachycardia-induced cardiomyopathy due to supraventricular arrhythmia. With the weight of convention, it is logical that concurrent ventricular systolic dysfunction and arrhythmia would be interpreted as a result of dilated cardiomyopathy. Therefore, it is necessary to wait until ventricular function improves after abolishing the arrhythmia to establish the definitive diagnosis. This condition may be more frequent than has been suspected up to now. In one study, in 12 of 14 patients with dilated cardiomyopathy and frequent premature beats, considerable improvement in left ventricular function was observed in 4 of the 5 patients who experienced a notable drug-induced decrease in ventricular ectopic activity. This suggests that some patients diagnosed with ventricular arrhythmia secondary to dilated cardiomyopathy may actually have had the opposite problem: primitive ventricular arrhythmia--frequent premature beats in this case--able to induce ventricular dysfunction in a healthy heart.
With regard to the causative mechanism, we cannot invoke tachycardia as the determinant pathogenic factor in our patients, since the mean 24-hour heart rate was only slightly elevated. Thus, we must look to other mechanisms to explain the ventricular dysfunction. One such factor, atrioventricular asynchrony, can reduce the atrial contribution to ventricular filling and produce an increase in pressure through the baroreceptors that could trigger neurohumoral mechanisms typical of heart failure. Anomalous ventricular activation, common in ventricular ectopy, makes diastolic mitral regurgitation, and mechanical asynchrony (both interventricular and intraventricular) possible during contraction. The mechanism we propose for our cases is that an intraventricular conduction disturbance together with atrioventricular asynchrony and mild tachycardia led to the development of left ventricular dysfunction.
CLINICAL IMPLICATIONS
In the guidelines of the Spanish Society of Cardiology,13 the indication for ablation in monomorphic ventricular premature beats is considered exceptional. The recommendation for ablation in repetitive ventricular tachycardia is contemplated in patients with symptoms and those who fail to respond to or cannot tolerate drug therapy; tachycardia-induced cardiomyopathy is not mentioned. Considering that ablation in these tachycardia patients has a success rate of nearly 80% and a complication rate similar to that of other more common origins,14 the indication for this procedure should be assessed when there is frequent monomorphic ventricular arrhythmia (premature beats, whether isolated or associated with repetitive tachycardia) together with apparently idiopathic left-ventricular dysfunction.
Correspondence: Dr. L. Tercedor Sánchez.
Servicio de Cardiología. Hospital Universitario Virgen de las Nieves.
Avda. de las Fuerzas Armadas, 2. 18014 Granada. España.
E-mail: luis.tercedor.sspa@juntadeandalucia.es