Keywords
INTRODUCTION
The development of de novo stenotic lesions in venous and arterial grafts in patients with coronary revascularization surgery has increased the need for repeat interventions, whether a new operation or a percutaneous coronary intervention (PCI) with angioplasty using a conventional balloon, atherectomy and/or stent placement.1 The first percutaneous transluminal coronary angioplasty in a saphenous vein graft was performed in 1980 and the use of PCIs has extended to internal mammary artery (IMA) grafts and to gastroepiploic artery grafts, with good results.2-4
Currently, the radial artery (RA) is favored as a free graft for coronary revascularization. The proximal anastomosis is generally made to the ascending aorta, although the RA can be used as a graft from other vessels with various techniques.5 In the present case, we describe a PCI to treat stenosis of a radial graft with a proximal T anastomosis to the left mammary artery.
CASE STUDY
A 59-year-old man, ex-smoker, with a history of hypertension, type 2 diabetes, and dyslipidemia, presented with unstable angina due to 3-vessel disease in 2000. At that time, he was treated with coronary revascularization surgery consisting of a double bypass: left IMA to the left anterior descending artery (LAD) and a radial artery T graft from the left IMA to the posterolateral branch of the LAD, without complications. In 2002, the patient again presented unstable angina. Coronary angiography showed a patent left IMA to the LAD and a severe focal lesion in the proximal third of the radial graft, which persisted after intracoronary injection of 200 µg of nitroglycerin to rule out vasospasm (Figure 1). A PCI through the IMA was decided to treat the radial graft stenosis. Using a femoral approach, a 6 Fr Z2 guide catheter (Medtronic-AVE, Santa Rosa, CA, USA) was advanced through the IMA. The lesion was crossed with a ChoICE® PT Extra Support guide wire (Boston Scientific Inc, Natick, MA, USA), predilatation was achieved with a 2.75x12-mm Crosssail Coronary Dilatation Catheter balloon (single inflation to 8 atmospheres) and elective implantation of a 2.75x15-mm Multi-link RX Zeta® stent (both from Guidant Corp., Santa Clara, CA, USA) was performed with good results (Figure 2). During stent placement, a small indentation was observed, which disappeared when the final pressure of 12 atmospheres had been reached. The procedure was performed under continuous intravenous nitroglycerine infusion because of RA sensitivity to vasospasm and with glycoprotein IIb/IIIa platelet inhibitors (Reopro®).6
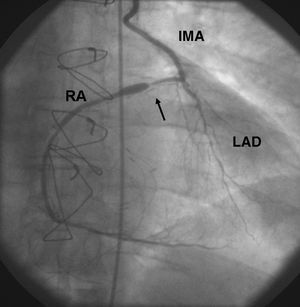
Figure 1. Posteroanterior coronary angiography through the IMA shows a severe proximal lesion in the radial T-graft with post-stenosis dilation (arrow). The LAD shows diffuse disease. IMA indicates left internal mammary artery; LAD, left anterior descending artery; RA, radial artery graft
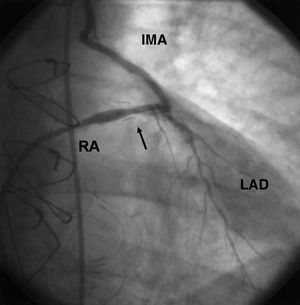
Figure 2. Post-procedure coronary angiography shows acceptable angiographic outcome (arrow). IMA indicates left internal mammary artery; LAD, left anterior descending artery; RA, radial artery graft.
At 6 months the patient presented symptoms of exertional angina. A new coronary angiography showed no restenosis of the proximal RA stent, but disclosed a new focal lesion in the middle third of the RA graft (Figure 3A). A PCI using the same technique as described above was performed and a 2.5x13-mm Pixel stent (Guidant Corp., Santa Clara, CA, USA) was successfully implanted in the graft without complications (Figure 3B).
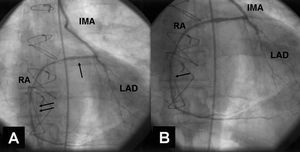
Figure 3. A: On coronary angiography at 6 months there is no restenosis of the dilated lesion in the proximal RA (arrow). A new lesion is seen in the middle third of the RA graft (double arrow). B: Final outcome following PCI for the de novo lesion (arrow). IMA indicates left internal mammary artery; LAD, left anterior descending artery; PCI, percutaneous coronary intervention; RA, radial artery graft.
At the 6-month clinical follow-up the patient remained asymptomatic. In addition we performed a non-invasive test to detect ischemia (single proton emission computed tomography [SPECT] with 99mTc for myocardial perfusion imaging), which was negative.
DISCUSSION
The radial artery was first used for coronary grafts at the start of the 1970s, but this practice was discontinued because of the high incidence of occlusion.7 The same working group "revived" radial grafts when RA bypasses were found to be patent on angiographic follow-up.8 Since then new strategies have been devised in the use of radial grafts, such as intraluminal dilatation with papaverin and administration of calcium channel blockers in the postoperative period. Now the long-term patency of radial grafts is higher than 80%.9
Several hypotheses have been put forward to explain the presence of obstructive lesions in radial grafts: focal or diffuse stenosis due to vasospasm, diffuse disease caused by competitive flow from native circulation, intimal hyperplasia resulting from the susceptibility of the middle layer to undergo ischemia, endothelial injury during manipulation of the graft, or technical problems during surgery.10,11 In the largest series of PCIs performed in radial grafts, it was found that two-thirds of cases of radial graft stenosis occur at the proximal and distal anastomoses. The same is true for IMA grafts, suggesting that the main origin of stenosis in these patients is related to technical difficulties.12 In our patient, stenosis was probably linked to intimal hyperplasia, based on the location of the lesion in the proximal third of the graft and the time window of more than 1 year between surgery and the onset of symptoms.
There are few reports of PCIs performed in radial grafts.12-14 Radial artery grafts, which are free and long, can be anastomosed to various vessels, although the proximal anastomosis is generally to the ascending aorta. In some patients the surgeon may prefer to create a radial artery bypass with proximal anastomosis to the left IMA, or suture the radial artery to a saphenous vein graft, the right IMA, another radial graft, the innominate artery, another coronary artery or, in minimally invasive surgery, to the descending aorta and the circumflex artery.5,15,16 When stenotic lesions occur in grafts in which the proximal anastomosis is not the ascending aorta, the PCI is performed with some variations on the original technique of dilatation for native arteries and other types of bypass, with more frequent use of stents as compared to procedures done in other arterial grafts, such as those using IMA.12,13 The limited current experience of PCI in radial grafts has indicated a success rate similar to that of IMA grafts, but with a comparatively higher incidence of restenosis, probably related to its use as an arterial bypass in a high percentage of patients with diabetes.12
This brief report describes a PCI in a RA bypass with a proximal T anastomosis to the left IMA, with initial success and no restenosis on angiographic follow-up. Nevertheless, a new procedure was required to treat a de novo lesion in the same graft. With the increased use of radial grafts for coronary revascularization, interventional cardiologists may encounter more cases requiring percutaneous procedures in this type of graft, with the proximal anastomosis outside the ascending aorta and the potential technical difficulties this may involve. In addition, because these grafts are used in diabetic patients and there is a potentially high risk of restenosis following PCI, the use of coated stents may play an important role in these procedures.17
Correspondence: Dr. A. Beloscar.
Rosers, 4 (Cala Romana). 43007 Tarragona. España.
E-mail: beloscar@wanadoo.es