Heart failure is a leading cause of morbidity and mortality in Western countries. Biventricular pacemakers have been used to treat heart failure since the 1990s.1 Over the last decade, randomized studies2,3 have demonstrated the benefit of cardiac resynchronization therapy (CRT) and helped to establish its indications. This therapy has been shown to increase survival and decrease hospitalizations in patients with heart failure, left ventricular (LV) dysfunction, and prolonged QRS, in particular in those with complete left bundle branch block.4 Unfortunately, a significant number of patients (30%-40%) have no response to CRT.5 This lack of response could be explained by inappropriate pacing site selection, suboptimal device programming, or absence of dyssynchronous basal LV contraction.5 Another limitation could be that pacing from a single LV point is incapable of generating a coordinated mechanical activation. Quadripolar electrodes would allow LV pacing from 2 points far enough apart to reduce activation time and LV dyssynchrony. A newly-marketed device (QUADRA ASSURA, St. Jude Medical) achieves multipoint LV pacing using a quadripolar lead. Compared with conventional CRT, initial results of multipoint pacing have shown an additional hemodynamic benefit.6 Our aim was to describe the first use of multipoint pacing in Spain, and evaluate its benefit over conventional CRT, using echocardiography.
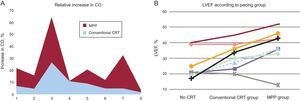
Following informed consent, 8 patients with indications for conventional CRT were enrolled and underwent device implantation between November 2013 and March 2014. A St. Jude Medical automatic implantable CRT-defibrillator was used, capable of pacing from multiple poles with the left ventricular lead. We compared baseline situation (no ventricular pacing), pacing from 1 LV point (conventional group), and pacing from 2 LV points (Multipoint Pacing [MPP] group). We used an anatomical configuration for multipoint pacing, from the distal and proximal poles of the LV lead (47 mm apart), with the goal of capturing the maximum myocardial mass possible. The device was programmed in AAI at 90 bpm (baseline group) and DDD at 90 bpm (conventional group and MPP group) to avoid the effect of cardiac frequency on cardiac output variation. Hemodynamic evaluation was performed using transthoracic echocardiography (IE33, Philips®) at least 1 month post-implant, by an echocardiographer blinded to the device program. The same operator repeated the measurements masked, to determine variability. Ejection fraction was calculated using the Simpson method, and cardiac output using the time-volume integral. Descriptive statistics were calculated. To determine intra-observer variability, an intraclass correlation coefficient was used. The Kolmogorov-Smirnov normality test was performed due to small sample size. Normally distributed variables were analyzed using ANOVA (with Bonferroni correction for multiple comparisons); those with non-normal distribution were compared using the Kruskal-Wallis test. We performed the statistical analysis using IBM SPSS Statistics 18, defining significance as a P-value of < 0.05. Patient characteristics are summarized in the Table. There were no complications during implantation, and no subsequent electrode dislocation. Echocardiographic calculations showed low variability (intra-class correlation coefficient 0.965). Seven patients (88%) showed hemodynamic improvement with resynchronization activation. Of those patients, 6 (86%) achieved greater improvement with multipoint pacing. Mean cardiac output (SD) was 4.8 L/min (0.8), 5.3 L/min (0.9), and 5.6 L/min (1.2) in the baseline, conventional and MPP groups, respectively. We studied the relative increase in cardiac output in the CRT patients, and found greater increases in the MPP group (16.7%), compared with the conventional group (10.4%), but the difference was not statistically significant (P = .2) (Figure A). Lastly, we studied the influence of multipoint pacing on LV ejection fraction, comparing the conventional, baseline and MPP groups [25.1% (3.2), 31.8% (3.0), and 37.8% (4.2) respectively]. Compared with the baseline group, the mean increase in LV ejection fraction was greater in the MPP group than in the conventional group: 12.7% (P=.055) vs 6.7% (P=.58) (Figure B).
Sample Characteristics (N=8)
| Women, % | 3 (37.5) |
| Age, mean (SD), y | 63 (8) |
| Underlying Heart Disease | |
| Dilated Cardiomyopathy | 7 (87.5) |
| Ischemic Cardiomyopathy | 1 (12.5) |
| CVRF | |
| HTN | 4 (50) |
| Dyslipidemia | 6 (75) |
| Diabetes mellitus | 4 (50) |
| Smoking | 4 (50) |
| LVEF, mean (SD), % | 22.5 (8) |
| Treatment | |
| Loop diuretics | 7 (87.5) |
| Beta-blockers | 6 (75) |
| ACE-I/ARB | 7 (87.5) |
| Aldosterone antagonists | 6 (75) |
| Anti-arrhythmics | 1 (12.5) |
| NYHA FC | |
| II | 2 (25) |
| III | 5 (62.5) |
| IV | 1 (12.5) |
| Baseline ECG rhythm | |
| Sinus rhythm | 7 (87.5) |
| Atrial fibrillation | 1 (12.5) |
| QRS width, mean (SD), ms | 162 (20) |
| Intraventricular Conduction | |
| CLBBB | 6 (75) |
| Nonspecific abnormalities | 1 (12.5) |
| Ventricular paced rhythm | 1 (12.5) |
ACE-I, angiotensin-converting enzyme inhibitor; ARB, angiotensin II receptor antagonist (blocker); CLBBB, complete left bundle branch block; CVRF, cardiovascular risk factor; ECG, electrocardiogram; LVEF, left ventricular ejection fraction; NYHA FC, New York Heart Association functional class; SD, standard deviation.
The values are expressed as N (%) or mean (standard deviation).
This prospective single-center study describes the first use of multipoint pacing in Spain. The hemodynamic data obtained from multipoint pacing are promising, because they show an improvement on the outcome of traditional resynchronization. Use of simultaneous stimulation from multiple points of the LV could lead to a reduction in the rate of non-responders and further improvement for those who do respond.
The main limitation of the study is the small sample size, and therefore further validation is required with larger-scale studies. Also, the study used acute hemodynamic data, so medium- and long-term follow-up is required to assess the clinical benefit of multipoint pacing.