Total anomalous pulmonary venous connection accounts for 1.5% of all congenital heart defects.1 Although the total postoperative mortality for total anomalous pulmonary venous connection has dropped to 10%, mortality can rise to 40% to 60% in patients with postoperative venous obstruction.1,2 This type of obstruction can occur in the anastomosis or in the pulmonary vein ostium.
This condition has been treated by balloon dilation or stent implantation in the pulmonary vein ostium,3,4 although sutureless surgery is now considered the treatment of choice.5
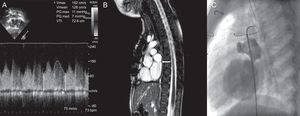
We describe a 4-year-old boy diagnosed at age 1 month with nonobstructive supradiaphragmatic total anomalous pulmonary venous connection, in which the pulmonary veins converged into a common chamber that drained into the innominate vein. Surgical repair was performed 2 months later, and the chamber was anastomosed to the left atrium roof. The postoperative period was unremarkable. After 2 years, echocardiography revealed a possible stenosis in the anastomosis (Figure 1A), which was later confirmed by magnetic resonance imaging (Figure 1B).
A: Echocardiography with pulsed Doppler in anastomosis, with loss of the biphasic pattern in pulmonary venous flow and a mean gradient of 7mmHg. B: Sagittal plane on cardiac magnetic resonance imaging; stenosis in the anastomosis, with a 6-mm diameter (arrow). C: Angiography (lateral view); dilated chamber connected to the left atrium roof through a stenosed orifice. Bpm, beats per minute; PG, pressure gradient; Vmax, maximum velocity; Vmean, mean velocity; VTI, velocity-time integral.
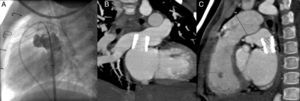
The patient underwent cardiac catheterization via a femoral vein approach and radiofrequency perforation of the interatrial septum. The pulmonary pressure was 27/15 (25) mmHg; aortic pressure, 63/40 (50) mmHg; pulmonary wedge pressure, 18mmHg; and chamber-to-left atrium gradient, 7mmHg. Angiography revealed dilatation of the pulmonary veins and chamber, with a diameter of 6.1mm in the anastomosis with the left atrium (Figure 1C). Angioplasty with a 15×25-mm Cristal Balloon catheter was unsuccessful and, therefore, a decision was made to implant a stent in the anastomosis. A 0.035-inch Amplatz Super-Stiff™ guidewire and a Mullins 9 Fr sheath were used to advance a 16-mm IntraStent® LD Mega™ fitted to a 14×35-mm BIB® (balloon-in-balloon) catheter, which was implanted in the anastomosis (Figure 2A). The gradient between the chamber and the left atrium dropped to 2mmHg. There were no complications, and acetylsalicylic acid and clopidogrel therapy was initiated.
During follow-up, echocardiography showed persistent dilatation of the pulmonary veins, with a slightly higher velocity, but the biphasic flow pattern of the pulmonary veins was maintained. Two years after the procedure, follow-up computed tomography angiography showed a possible stenosis at the stent angle with the right pulmonary veins (Figures 2B and C) and, therefore, cardiac catheterization was repeated. The mean pulmonary pressure was 15mmHg; the mean aortic pressure, 54mmHg; and the pulmonary wedge pressure, 11mmHg, equal to the left ventricular end-diastolic pressure. Transesophageal echocardiography showed laminar flow through the stent and stent cells, but no endothelial proliferation. Because no stenosis was observed, it was decided to take a wait-and-see approach and maintain long-term dual antiplatelet therapy to avoid the risk of thromboembolism.
Postoperative venous obstruction following surgical repair of total anomalous pulmonary venous connection is a severe complication, particularly when it presents early, is bilateral, and affects the ostium of the pulmonary veins.1,2 In these cases, endothelial proliferation narrows the pulmonary vein lumen and usually recurs after stent implantation or classic surgical repair.2–4 In the case of our patient, the prognosis is good because the postoperative venous obstruction developed 2 years after the surgery and only affected the anastomosis. Hence, we believed that the implantation of a large stent at some distance from the pulmonary vein ostium would not favor endothelial proliferation. The stent lumen and lateral cells have remained patent during the patient's clinical course, which supports our hypothesis.
To our knowledge, this is the first case of the successful implantation of a stent to resolve postoperative venous obstruction in the anastomosis. The follow-up period is short, and in-stent stenosis could develop in the future. However, the possibility of dilation up to 22mm provides ample margin for surgery, and if surgery is deemed necessary, stent removal should pose no difficulties.
In conclusion, we believe that stent implantation may prevent repeat surgery in postoperative anastomotic venous obstructions after total anomalous pulmonary venous connection surgery, at least in the short- and medium-term.