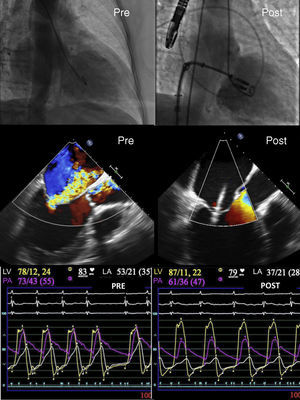
Treatment of functional mitral regurgitation (FMR) is currently a challenge for the clinician. The association of severe FMR and heart failure significantly increases mortality. Although the risk of surgical valve repair is often high in patients with these 2 conditions, the valve anatomy rarely impedes MitraClip implantation. Nonetheless, even though more than 35 000 MitraClip devices have been implanted worldwide, FMR is not an accepted indication due to a lack of randomized studies in these patients. Only 1 study has included patients with mitral regurgitation of largely organic origin. On the basis of the results of that study, only patients with organic mitral regurgitation are accepted for implantation of the MitraClip device as an alternative to surgery. The European clinical practice guidelines do not give a strong recommendation for this indication (IIB with level of evidence C). However, in Europe, FMR has been the main reason for MitraClip implantation. According to data provided by the manufacturer, of 25 000 patients treated with this technique up to 2016, 65% had FMR. In Spain, the proportion is even higher (78%). This large number of procedures is due to the good outcomes obtained in observational studies (Figure). Two large European registries, ACCESS-EU1 and TRAMI,2 which included 567 and 749 patients, respectively, with high surgical risk reported incidences of FMR of 77% and 71%. The MitraClip was effective in these patients, with low rates of mortality and adverse effects.
Similar results were obtained in a Spanish registry that included patients with a high incidence of FMR (85%) and high surgical risk.3 The procedure was safe, reduced mitral regurgitation, and improved patients’ functional capacity.
In a nonrandomized study, Giannini et al.4 reported decreased mortality with the use of this device vs medical treatment in patients with FMR.
Finally, a recent meta-analysis evaluated all available experience with MitraClip in FMR.5 The study included 875 patients from 9 observational studies. The meta-analysis concluded that the MitraClip is an effective strategy for patients with heart failure and severe mitral regurgitation. The procedure significantly improves functional class and cardiac remodeling.
Although the efficacy of the device in reducing the degree of mitral regurgitation is beyond question (Figure), mortality in patients with severe mitral regurgitation remains high. Azzalini et al.6 reported a probability of survival at 3 years of close to 30% and an ejection fraction of less than 27% in patients with FMR treated with MitraClip. For this reason, some clinicians have considered mitral valve repair at earlier stages of the disease and have sought treatment alternatives for patients with highly deteriorated function.
Accordingly, the criteria for device implantation vary for different hospitals and randomized studies are required for a well-established indication. In patients with less advanced disease and better ventricular function, the clinical outcomes of the technique would likely be better but, at the same time, the natural course of the disease with medical treatment would be more benign; therefore, randomized trials are needed. Four large multicenter studies have been designed (Table). The first is the COAPT study (NCT01626079), which plans to randomize 430 patients with FMR 1:1 to MitraClip or medical treatment. The second is the RESHAPE-HF2 study (NCT02444338), which will enroll 800 patients with FMR, randomized 1:1 to optimal medical treatment or MitraClip. The third, also currently ongoing, is the MITRA-FR study (NCT01920698), which will enroll 288 patients with a similar primary outcome measure to the previous studies. Finally, the MATTERHORN study (NCT02371512) will randomize 210 patients to mitral repair surgery or a MitraClip procedure (Table).
Ongoing Randomized Studies of the MitraClip and Functional Mitral Regurgitation.
| Study | COAPT | RESHAPE-HF2 | MITRA-FR | MATTERHORN |
|---|---|---|---|---|
| Patients/hospitals, n | 430/75 | 800/50 | 288/18 | 210/1 |
| Control group | Medical treatment | Medical treatment | Medical treatment | Mitral repair surgery |
| FMR severity | ≥ 3+ (EROA ≥ 30 mm2 or Rvol > 45 mL according to CLE) | ≥ 3+ (EROA ≥ 30 mm2 or Rvol > 45 mL according to CLE) | Severe (EROA > 20 mm2 + Rvol > 30 mL) | Severe (according to guidelines) |
| NHYA functional class | II, III, or ambulatory IV | III or ambulatory IV | II-IV | III-IV |
| Other inclusion criteria | Admission for HF in last 12 mo or BNP ≥ 300 pg/mL or NT-proBNP ≥ 1500 pg/mL in the last 12 mo. No indication for surgery in the center | Admission for HF in last 12 mo or BNP ≥ 350 pg/mL or NT-proBNP ≥ 1400 pg/mL in 90 d. Not apt for surgery | Admission for HF in last 12 mo. Not apt for surgery | Clinically symptomatic FMR. High surgical risk, but acceptable for surgery |
| LVEF | ≥ 20%-≥ 50% | ≥ 15%-≥ 40% | ≥ 15%-≥ 40% | ≥ 20%-≥ 45% |
| Ventricular size | LVEDD ≥ 70 mm | LVEDD ≥ 55 mm | — | — |
| Primary outcome measure | Admission for HF in last 12 mo | Death or need for admission for HF in last 12 mo | Death or need for admission for HF in last 12 mo | Death, readmission for HF, repeat procedure, VSD implantation, or stroke in 12 mo |
| Follow-up | 5 y | 2 y | 2 y | 1 y |
BNP, brain natriuretic peptide; ECL, echocardiography core laboratory; EROA, effective regurgitant orifice area; FMR, functional mitral regurgitation; HF, heart failure; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; Rvol, regurgitation volume; VSD, ventricular support device.
In conclusion, the best of 2016 with regard to the MitraClip in the treatment of FMR will actually come in 2017, with the availability of the results of the ongoing studies. The device effectively reduces mitral regurgitation and improves clinical and hemodynamic parameters (Figure). With the currently available results, a change is already needed to the guidelines and, if the ongoing studies are positive, a recommendation will no doubt be established with a higher degree of evidence.
CONFLICTS OF INTERESTM. Pan has received minor payments from Abbott for presentations.