The severity of preoperative right ventricular dysfunction contributes to early and long-term postoperative outcomes of heart valve surgery; therefore, patients with abnormal right ventricular contractility or morphology are considered to be at high perioperative risk.1,2 EuroSCORE I and II underestimate the risk of these patients because right ventricular chamber parameters are not included in these risk scores. Several studies have demonstrated the benefit of preoperative levosimendan administration in patients with ventricular dysfunction.3 Preoperative levosimendan administration4 improved myocardial function in the early postoperative period and its early use produced significantly more benefits than late administration.3,4 The preoperative use of levosimedan has been evaluated in patients with left ventricular dysfunction4,5 undergoing cardiac or noncardiac surgery but not in patients with right ventricular dysfunction. We report a case series of patients with right ventricular dysfunction who were administered levosimendan before heart valve replacement surgery to prevent right ventricular failure as the primary target.
Preoperative levosimendan was prescribed to 9 patients with tricuspid valve disease and/or right ventricular dysfunction. Our aim was to determine whether levosimendan improved hemodynamic parameters, inotropic support, and renal function within the first 48 h of the postoperative period. The indication of preoperative levosimendan was based on tricuspid annular plane systolic excursion (TAPSE) <15 or the presence of moderate or severe right ventricular dilatation (moderate, 33-39mm; severe >39mm) on Doppler echocardiography. After the 9 patients had provided informed consent, levosimendan was administered before surgery to optimize cardiac function (all the patients were in functional class IV/IV) as continuous intravenous infusion, without previous bolus injection, at a rate of 0.15-0.10µg/kg/min until a cumulative dose of 12.5mg was reached. Standard routine monitoring was used (electrocardiogram, noninvasive blood pressure measurement, urine output and body temperature). None of the patients required cessation of infusion due to adverse effects. Central venous pressure was measured before the patient entered the operating room (12-16mmHg). All the patients underwent heart valve replacement surgery (Table).
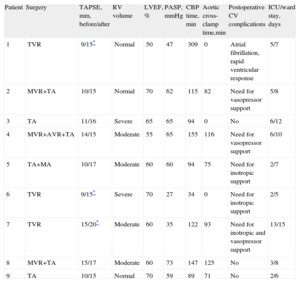
Preoperative, Operative, and Postoperative Data of the 9 Patients
| Patient | Surgery | TAPSE, mm, before/after | RV volume | LVEF, % | PASP, mmHg | CBP time, min | Aortic cross-clamp time,min | Postoperative CV complications | ICU/ward stay, days |
| 1 | TVR | 9/15* | Normal | 50 | 47 | 309 | 0 | Atrial fibrillation, rapid ventricular response | 5/7 |
| 2 | MVR+TA | 10/15 | Normal | 70 | 62 | 115 | 82 | Need for vasopressor support | 5/8 |
| 3 | TA | 11/16 | Severe | 65 | 65 | 94 | 0 | No | 6/12 |
| 4 | MVR+AVR+TA | 14/15 | Moderate | 55 | 65 | 155 | 116 | Need for vasopressor support | 6/10 |
| 5 | TA+MA | 10/17 | Moderate | 60 | 60 | 94 | 75 | Need for inotropic support | 2/7 |
| 6 | TVR | 9/15* | Severe | 70 | 27 | 34 | 0 | Need for inotropic support | 2/5 |
| 7 | TVR | 15/20* | Moderate | 60 | 35 | 122 | 93 | Need for inotropic and vasopressor support | 13/15 |
| 8 | MVR+TA | 15/17 | Moderate | 60 | 73 | 147 | 125 | No | 3/8 |
| 9 | TA | 10/15 | Normal | 70 | 59 | 89 | 71 | No | 2/6 |
AVR, aortic valve replacement; CBP, cardiopulmonary bypass; CV, cardiovascular; ICU, intensive care unit; LVEF, left ventricular ejection fraction; MA, mitral valve annuloplasty; MVR, mitral valve replacement; PASP, pulmonary artery systolic pressure; RV, right ventricular; TA, tricuspid valve annuloplasty; TAPSE, tricuspid annular plane systolic excursion; TVR, tricuspid valve replacement.
Forty-eight hours after levosimendan administration, Doppler echocardiography showed improvement of TAPSE in all patients except one.
In patients with a tricuspid valve prosthesis, both TAPSE and right ventricular ejection fraction improved.
Regarding the use of vasopressor and inotropic agents, 3 patients required low-dose norepinephrine, which was stopped in 2 of them after extubation (within 12 h after surgery). Another 3 patients required inotropic support with dobutamine or epinephrine during the first day after surgery (which was withdrawn before they underwent Doppler echocardiography); in 1 of these patients, treatment was continued during the second day after surgery. During the first 48 h, in 8 patients creatinine levels and urine output remained within normal values under furosemide at a mean dose of 30 mg/day; 1 of the patients developed kidney failure and needed renal replacement therapy.
We have reported the largest case series of patients who underwent levosimendan infusion to improve right ventricular function before heart valve replacement surgery.
In 8 of these nine patients, right ventricular contractility was impaired before surgery and improved after surgery with preserved renal function. Our patients belonged to a group commonly associated with longer stay in intensive care units and with greater mortality than other types of patients. Case series have no control group or other treatment group and consequently, given our results, further studies are warranted. Finally, as there were no complications associated with preoperative levosimendan infusion, we may assume that this technique is safe and could benefit patients with high perioperative risk, such as those with right ventricular dysfunction undergoing heart valve replacement surgery.
.