Extracorporeal membrane oxygenation (ECMO) has proven effective for providing respiratory and circulatory support in patients with refractory cardiogenic shock or severe respiratory failure.1 Formerly, this therapy was limited to certain tertiary hospitals performing transplants, but over the last few years, many centers have initiated ECMO programs. The development of new, more compact ECMO systems has enabled transport of critically ill patients in relative comfort, and in safer and more favorable hemodynamic conditions. The creation of mobile units with trained professionals that can provide on-site care, with stabilization and subsequent transfer to a specialized center, offers these patients a chance for survival.2
In October 2013, Hospital Universitario de Salamanca launched its ECMO program, which was extended to a mobile ECMO program starting in June 2014. We performed a retrospective analysis of patients hospitalized with ECMO support in our center. Since its implementation, 9 patients have undergone interhospital transfer with ECMO. The aim of this report was to evaluate the feasibility and safety of an interhospital transfer program using ECMO for critically ill patients. We describe the logistic problems, indications, and outcome of our series.
Our mobile ECMO program includes 2 scenarios. The first is transfer of a patient receiving ECMO from our hospital to a reference hospital for heart or lung transplant. In this case, the attending team is composed of a perfusionist, a physician, and a nurse with extensive experience of ECMO. The second possibility is to transport a team to a center without an ECMO program, provide the care needed, and then transfer the patient to our center. In this scenario, the team additionally includes a physician experienced in cannulation, who could be a cardiologist or surgeon, depending on the type of support required. The response time in our program is < 90 minutes from the decision to mobilize the team to its departure, and the service is permanently available, 365 days a year.
The vehicle used is an advanced life support ambulance, equipped with a CARDIOHELP system (MAQUET, Cardiopulmonary AG, Germany). Peripheral cannulation through femoral access is performed according to the Seldinger technique, using 17 to 21 Fr arterial cannulas and 21 to 29 Fr venous cannulas. Cannulation of the superficial femoral artery for distal limb perfusion is carried out using a 6 to 7 Fr catheter under Doppler ultrasound guidance. Implantation is controlled by transesophageal echocardiography. During transfer, patients are under sedation and pain medication and are connected to mechanical ventilation.
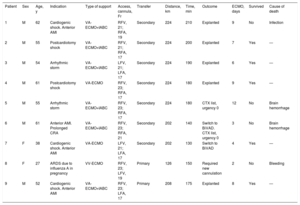
From October 2013 to August 2016, 9 critically ill patients (7 men and 2 women, mean age 51.7 ± 11.7 [range 27-62] years) required transfer on ECMO. In 2 cases, the team went to other centers for ECMO implantation and in the remaining cases, the patients were transferred from our hospital to centers performing cardiac transplantation. The patients’ demographic characteristics and the reasons why circulatory/respiratory support was needed are summarized in Table. The most frequent indication was cardiogenic shock due to myocardial infarction in 4 patients (44.4%). Eight patients required venoarterial ECMO support and 6 of them additionally needed intra-aortic balloon counterpulsation therapy. In 1 patient, venovenous ECMO was implanted for acute respiratory distress syndrome. Percutaneous cannulation was performed in 7 patients, and surgical placement in 2 patients with postcardiotomy shock. The mean distance travelled was 206.4 ± 31.7 km and the mean transport time was 172.8 ± 27.3minutes. The patients had no complications, transport-related morbidity or mortality, or device-related logistic/technical complications. The mean time on support was 6.7 (2-12) days. In-hospital survival was 55.6%, and all surviving patients were alive at the time of writing. The causes of death were bleeding complications (75.0%) and infections (25.0%). Weaning from ECMO was achieved in 55.6% of patients, although 1 died 2 weeks later due to sepsis secondary to pneumonia. Two patients (cases 6 and 7) who required a longer time on support were switched to a long-term biventricular system; only patient 7 survived. Two of the 4 patients who died were on the cardiac transplant waiting list (urgency 0); their deaths were due to bleeding complications.
Demographic Characteristics, and Transport- and Outcome-related Variables in Included Patients
| Patient | Sex | Age, y | Indication | Type of support | Access, cannula, Fr | Transfer | Distance, km | Time, min | Outcome | ECMO, days | Survived | Cause of death |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M | 62 | Cardiogenic shock. Anterior AMI | VA-ECMO+IABC | RFV, 21; RFA, 19 | Secondary | 224 | 210 | Explanted | 9 | No | Infection |
| 2 | M | 55 | Postcardiotomy shock | VA-ECMO+IABC | RFV, 21; RFA, 17 | Secondary | 224 | 200 | Explanted | 7 | Yes | — |
| 3 | M | 54 | Arrhythmic storm | VA-ECMO+IABC | LFV, 21; LFA, 17 | Secondary | 224 | 190 | Explanted | 6 | Yes | — |
| 4 | M | 61 | Postcardiotomy shock | VA-ECMO | RFV, 23; RFA, 17 | Secondary | 224 | 180 | Explanted | 9 | Yes | — |
| 5 | M | 55 | Arrhythmic storm | VA-ECMO+IABC | RFV, 23; RFA, 17 | Secondary | 224 | 180 | CTX list, urgency 0 | 12 | No | Brain hemorrhage |
| 6 | M | 61 | Anterior AMI. Prolonged CRA | VA-ECMO+IABC | RFV, 23; RFA, 21 | Secondary | 202 | 140 | Switch to BiVAD. CTX list, urgency 0 | 3 | No | Brain hemorrhage |
| 7 | F | 38 | Cardiogenic shock. Anterior AMI | VA-ECMO | LFV, 21; LFA, 17 | Secondary | 202 | 130 | Switch to BiVAD | 4 | Yes | — |
| 8 | F | 27 | ARDS due to influenza A in pregnancy | VV-ECMO | RFV, 23; LFV, 19 | Primary | 126 | 150 | Required new cannulation | 2 | No | Bleeding |
| 9 | M | 52 | Cardiogenic shock. Anterior AMI | VA-ECMO+IABC | RFV, 23; LFA, 17 | Primary | 208 | 175 | Explanted | 8 | Yes | — |
AMI, acute myocardial infarction; ARDS, acute respiratory distress syndrome; BiVAD, biventricular assist device; CRA, cardiorespiratory arrest; CTX, cardiac transplant; F, female; IABC, intra-aortic balloon counterpulsation; LFA, left femoral artery; LFV, left femoral vein; M, male; RFA, right femoral artery; RFV, right femoral vein; VA-ECMO, venoarterial extracorporeal membrane oxygenation; VV-ECMO, venovenous extracorporeal membrane oxygenation.
A percentage of critically ill patients do not respond to conventional treatment and must be transported to centers offering more advanced specialized care. Often, these patients are hemodynamically unstable, and their transfer implies a high risk.3 In these cases, ECMO provides respiratory and circulatory support that facilitates transfer and improves prognosis. Several articles have reported data on interhospital transfer of patients on ECMO,2,3 but there is little information in this line from Spain. Groups that manage large numbers of patients have indicated that there are few complications during transport, and mortality rates are low, around 0.5%.3 No complications occurred in our series during transfer.
There is no standard definition of an “ECMO center”, but various publications have cited a minimum of 20 cases per year to complete an adequate learning curve and achieve a decrease in mortality after 30 patients per year.4
Our increasing experience in the use of ECMO has been extended to include applications in other less common indications, as previously reported in Revista Española de Cardiología.5 The present report describes our experience with interhospital transfer of patients under ECMO support. Our results show that it is a safe and effective option for transporting unstable patients. Widespread use of this resource could facilitate the creation of health care networks for the treatment of cardiogenic shock in Spain, with the aim of centralizing experience of this option and improving patient prognosis.