Researchers are increasingly interested in the study of complex atheromatosis of the aortic arch (plaque thickness ≥ 4mm, ulceration, or mobile elements) by transesophageal echocardiography because such lesions are a potential major origin of emboli in different types of cryptogenic cerebral ischemia. The prevalence of complex AA was found to be approximately 7.6%.1 Several studies confirm the higher frequency of complex aortic atheromatosis (AA) among patients with ischemic stroke or transient ischemic attack (TIA) compared with healthy controls. However, we lack in-depth knowledge of the prevalence of complex AA associated with these different clinical presentations of an ischemic event. The objective of this study was therefore to assess the possible differences between stroke and TIA.
The study was a retrospective observational study of 100 consecutive patients attended between 2004 and 2011 (50 with stroke and 50 with TIA). These patients had been referred for transesophageal echocardiography due to cryptogenic cerebrovascular accident after a complete diagnostic work-up that included electrocardiogram, chest radiograph, brain computed tomography/magnetic resonance imaging, Doppler ultrasound of the supra-aortic arteries (in 3 patients in the stroke group and in 2 in the TIA group of mild carotid stenosis), brain magnetic resonance angiography, blood tests to rule out hypercoagulation, and conventional transthoracic echocardiography. The 2 types of event were differentiated according to whether infarcted tissue was detected in the central nervous system by the imaging techniques recommended at the time.2 Patients with intracerebral or subarachnoid bleeding and evidence of atrial fibrillation and those younger than 18 years were excluded.
Of the 100 patients included initially, 17 were excluded (8 [16%] in the TIA group and 9 [18%] in the stroke group) due to patent foramen ovale that was not detected by conventional transthoracic echocardiography. Therefore, the final sample comprised 83 patients. None of the patients had thrombi in the left atrial appendage.
Aortic examinations were undertaken according to the current recommendations of the European Society for Cardiology using a multiplanar transesophageal probe (2.9-7.0MHz) connected to a Vivid7 (GE, Vingmed, Horten, Norway) echocardiography unit. Complex AA was defined as maximum plaque thickness ≥ 4mm and presence of ulcerations or mobile elements within the plaques.
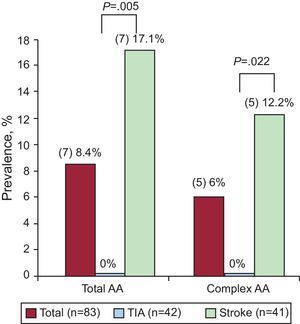
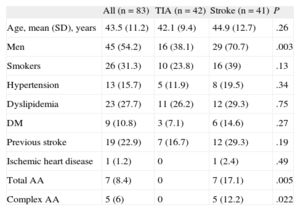
The clinical characteristics of the groups are shown in the Table. Stroke patients were older than TIA patients and had a worse cardiovascular risk profile (classic risk factors), although these differences were not significant. The prevalence of total and complex AA in the stroke group was significantly greater than in the TIA group (17.1% vs 0%;P=.005; and 12.2% vs 0%; P=.022, respectively), as shown in the Figure.
Clinical Characteristics of the Sample
| All (n = 83) | TIA (n = 42) | Stroke (n = 41) | P | |
| Age, mean (SD), years | 43.5 (11.2) | 42.1 (9.4) | 44.9 (12.7) | .26 |
| Men | 45 (54.2) | 16 (38.1) | 29 (70.7) | .003 |
| Smokers | 26 (31.3) | 10 (23.8) | 16 (39) | .13 |
| Hypertension | 13 (15.7) | 5 (11.9) | 8 (19.5) | .34 |
| Dyslipidemia | 23 (27.7) | 11 (26.2) | 12 (29.3) | .75 |
| DM | 9 (10.8) | 3 (7.1) | 6 (14.6) | .27 |
| Previous stroke | 19 (22.9) | 7 (16.7) | 12 (29.3) | .19 |
| Ischemic heart disease | 1 (1.2) | 0 | 1 (2.4) | .49 |
| Total AA | 7 (8.4) | 0 | 7 (17.1) | .005 |
| Complex AA | 5 (6) | 0 | 5 (12.2) | .022 |
AA, aortic atheromatosis; DM, diabetes mellitus; SD, standard deviation; TIA, transient ischemic attack.
Unless otherwise indicated, the data are expressed as No. (%).
Of the 5 stroke patients with complex AA, 3 (60%) had a plaque thickness > 4mm and 2 (40%) had mobile elements within the plaque. Oral anticoagulation was indicated in these patients.
Some studies have shown that complex thoracic aortic plaques, whether detected by transesophageal echocardiography or during autopsy, are much more frequent in patients with stroke than in controls.3 Although complex AA has not been associated with an increase in the primary risk of ischemic stroke in prospective studies, its presence has been considered a risk factor for recurrence.4
Very few studies have investigated whether there is a difference in AA prevalence between patients with ischemic cerebral stroke and TIA. In a retrospective study of 519 patients with complex AA, Tunick et al5 found that 50% had stroke, 35% TIA, and 14% peripheral embolism.
Aortic atherosclerotic plaques are considered a manifestation of systemic atherosclerosis and associations have been found with classic cardiovascular risk factors, old age, and coronary artery disease.6 In the present study, the findings could be partly explained by the greater cardiovascular risk, the greater percentage of men, and slightly older age in the stroke group compared to the TIA group. However, certain TIA episodes have been linked to fluctuation in cerebral artery flow, while atheroembolic mechanisms are not involved as in stroke.
Despite its small size, the study is important because it shows that patients with ischemic cerebral infarction of unknown origin have a higher prevalence of complex AA than those with TIA. Indeed, no cases of complex AA were found in this latter group. The most relevant conclusion probably pertains to the possible therapeutic and prognostic implications, given the indication of oral anticoagulation in patients at greatest risk of embolism (mobile elements adhering to the plaque) and the risk of recurrence of stroke.