Keywords
INTRODUCTION
Coronary artery perforation is an infrequent complication of percutaneous coronary interventions (PCIs) (0.2%-3%, depending on the series) that can have a poor prognosis because of the risk of cardiac tamponade, need for surgery, pseudoaneurysm formation, development of arrhythmia, and ultimate death.1-4
In 1994 Ellis et al1 proposed a prognosis-related classification of the degrees of coronary perforation following angioplasty. Later, in some reported cases of perforation caused by intracoronary guidewires, it was seen that the condition could progress to cardiac tamponade even though there was no contrast extravasation during the procedure.5,6
In the present study, we examine the incidence of coronary artery perforations caused by intracoronary guidewires, and evaluate the factors that can predict the development of this complication. We describe how this complication was managed and its short-term prognosis.
METHODS
A retrospective observational study was conducted, involving a review of medical records and catheterization laboratory data related to all consecutive patients who underwent PCIs from October 2000 to October 2008 at our hospital.
The variables assessed included classic cardiovascular risk factors, myocardial infarction, previous revascularization by coronary artery bypass surgery (CABG) or PCI, ventricular function, and pharmacological treatment, as well as technical factors, such as number of diseased vessels, severity of lesions, types of stents and guidewires used, presence of coronary calcifications and tortuosity, periprocedure medication, and the initial TIMI (thrombolysis in myocardial infarction) flow.
Definitions
Definitions used in the study are those that follow. Cardiac tamponade: systolic arterial pressure ≤90 mm Hg with paradoxical pulse and/ or pericardial effusion with echocardiographic signs of hemodynamic deterioration.7 Type A, B, or C coronary lesions, based on the criteria of the American College of Cardiology/American Heart Association (ACC/AMA).8 Coronary occlusion: absence of anterograde coronary flow (TIMI 0),9 which was considered to be chronic in cases of more than 3 months' evolution.10 Multivessel revascularization: revascularization of at least 2 different coronary arteries or the left main. Hydrophilic guidewires: guidewires with a polymer-coated tip, which facilitates navigation in tortuous vessels and severely stenotic coronary lesions; in the present study, the hydrophilic guidewires used were Pilot 50, Pilot 150, and Whisper (Abbott-Cardiovascular, Illinois, United States).11 Coronary artery perforation: extravasation of contrast to the pericardial space or intracardial cavity due to rupture of an epicardial artery, or pericardial effusion and positioning of the intracoronary guidewire outside the arterial bed.
Outcome Variables
The study outcome variables were coronary artery perforation caused by intracoronary guidewire, immediate or delayed (outside the revascularization laboratory) cardiac tamponade, early revascularization requirement (urgent CABG or PCI), and in-hospital death.
The statistical analysis was carried out with SPSS. Continuous variables were analyzed for homogeneity of variance and compared with the Student t test; results are expressed as the mean (SD). Categorical variables were compared with the Mann-Whitney test; results are expressed as absolute numbers (%). Statistical significance was set at a P value of <.05. Linear logistic regression analysis was performed with significant variables in the univariate analysis, and results are presented as the odds ratio (OR) adjusted by the 95% confidence interval (CI).
RESULTS
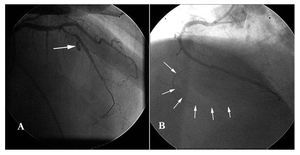
A total of 4353 consecutive PCI procedures to treat 6994 lesions were carried out during the study period. Thirty coronary perforations were detected, and 15 of them (0.35% of all PCIs) had been caused by an intracoronary guidewire (Figure 1).
Figure 1. A: coronary artery rupture following balloon dilation, with extravasation of contrast into the pericardial space. B: intracoronary guidewire perforation following advancement beyond the arterial vascular bed.
Variables related to guidewire perforation were the presence of more than 1 lesion per vessel (P=.016), number of diseased vessels (P=.005), complex lesions (P=.002), percentage of stenosis (P<.001), initial TIMI flow 0 (P<.001), use of more than 1 guidewire (P<.001), use of hydrophilic guidewires (P<.001) and treatment of chronic occlusions (P<.001) (Table 1). There was no relationship between the degree of urgency or clinical indication for the procedure and occurrence of guidewire perforation.
In the multivariate analysis, the use of hydrophilic guidewires (OR=2.33; 95% CI, 1.34-4.05) and treatment of chronic occlusions (OR=3.31; 95% CI, 1.05-10.46) were independently related to guidewire perforation.
In 8 patients, the diagnosis of coronary perforation was established by the presence of chest discomfort during the procedure or by clear visualization of the guidewire outside the vascular space; the clinical course progressed without complications in all cases. Symptoms onset in the remaining 7 patients consisted of varying degrees of subacute cardiac tamponade, and the diagnosis of coronary perforation was made a posteriori. Three of these 7, 3 patients presented nonspecific chest discomfort some hours before the diagnosis; in another 3, tamponade occurred after 24 hours, and in 1 patient, tamponade presented at 3 days postprocedure. In all patients, the diagnosis was confirmed by echocardiography and pericardiocentesis was performed, with a successful outcome in 3 patients. The 4 remaining patients required surgical drainage, which resulted in a favorable evolution in all cases (3 of them had acute myocardial infarction, treated with primary angioplasty, and free wall rupture during surgery was ruled out). Protamine was administered in all cases, and anti-GPIIb/IIIa perfusion was discontinued in patients receiving this treatment.
The development of cardiac tamponade was related to the number of coronary guidewires used (P=.039) and abciximab administration during the procedure (P=.016) (Table 2).
None of the patients required urgent CABG, and there were no deaths related to the procedure or any other cause.
DISCUSSION
In our setting, coronary artery perforation related to PCI was an uncommon complication, and in half the cases, the guidewire had gone beyond the vascular bed, without major rupture of the epicardial artery. The coronary guidewire perforations occurring in our patients were type 1, as described by Ellis et al.1
However, in contrast to the results of Ellis's study and another by Ramana et al,12 cardiac tamponade occurred in half the patients. This complication was related to the use of anti-GPIIb/IIIa, as has been reported by Gunning et al13 and Javaid et al15 in patients with vessel rupture. Other authors, such as Dippel et al14 and Ramana et al12 did not find this relationship. The high incidence of cardiac tamponade in the present study might be due to the fact that it was the symptoms of this condition that led to the diagnosis of perforation. It is likely that most cases of guidewire perforation go undetected because don't cause symptoms.
In half the patients, there were no initial symptoms or very nonspecific discomfort. In our opinion, it is particularly relevant that the symptoms of tamponade occurred subacutely (up to 3 days following the procedure). It may be that the passage of blood to the pericardium took place very slowly because of the small perforation produced by the guidewire. Thus, coronary artery guidewire perforation with associated cardiac tamponade should be suspected in all patients who present hypotension or chest pain following a PCI. An emergency echocardiogram can be very useful in these patients.
The treatment for coronary artery guidewire perforation has not been well defined. Anticoagulation agents were suspended in all patients in our series, and their effects were reversed by protamine administration.5,16 Anti-GPIIb/IIIa administration was interrupted in patients receiving this treatment and platelets were transfused. Double antiplatelet therapy was not suspended in any case.
Limitations
This is a retrospective study and only a small number of patients experienced the complication being investigated. Distal perforations may have been underdiagnosed because they can go unnoticed during angioplasty and developed no complications.
Lastly, protocol for the management of coronary perforation was left to the discretion of the interventional cardiologist.
CONCLUSIONS
Coronary artery guidewire perforation during percutaneous procedures is an uncommon complication that can sometimes have poor prognosis. In our series, perforation was related to multivessel disease, the degree of stenosis, the treatment of chronic occlusions, the use of hydrophilic guidewires, and the number of guidewires. The number of coronary guidewires used and the use of anti-GPIIb/IIIa predisposed to cardiac tamponade, which occurred subacutely and required pericardiocentesis or surgical drainage. In our series, there were no in-hospital deaths due to this serious complication.
Correspondence: Dr. E. Fernández-Nofrerías.
Sección de Hemodinámica del Servicio de Cardiología. Hospital Universitari Germans Trias i Pujol.
Ctra Canyet, s/n. 08916 Badalona. Barcelona. España.
E-mail: enofre@hotmail.com
Received December 29, 2008.
Accepted for publication July 20, 2009.