Keywords
INTRODUCTION
Surgical repair of patent ductus arteriosus (PDA) in adults is performed by left lateral thoracotomy, but may require a midline sternotomy, extracorporeal circulation, aortic cross-clamping, and other measures, with recovery in an intensive care unit. Surgical risk may be further increased by anatomic and histologic changes in the ductus, including calcification, aneurysm, diverticulum, shortening, and friability1,2 and, therefore, percutaneous occlusion as a first-line treatment is a valid option. If left untreated, PDA can foster the development of infective endarteritis, Eisenmenger syndrome, and other conditions, with an annual mortality rate of 1.8%.3 We report our experience with percutaneous treatment of PDA in adults using 3 different types of devices.
METHODS
A cross-sectional, retrospective, descriptive study was carried out between October 1992 and August 2008, including 23 patients (17.4% men). All patients were referred to our department for isolated PDA diagnosed by clinical symptoms, echocardiography and, in some cases, catheterization. Median patient age was 24 years (range, 16-75) and median weight was 57 kg (range, 52-80).
PDA repair was undertaken with the Rashkind-PDA (RAS) umbrella device in 9 patients; the cone-shaped Amplatzer Duct-Occluder (ADO) in 12, and the Nit-Occlud coil (NOc) in 2. All patients were followed up by clinical examination, radiologic study, and echocardiography.
Written informed consent was obtained for participation. In keeping with the protocol for these patients, general anesthesia was administered, introducer sheaths were positioned in the femoral artery and vein, and 5000 U of sodium heparin and intravenous antibiotic prophylaxis were administered. A single device, advanced from the femoral vein, was implanted in all patients. A hemodynamic study was performed, followed by thoracic aortography (right cephalic oblique and/or lateral projection using a 5-French pigtail catheter), in which the aortic and pulmonary artery diameters and the PDA length were measured. RAS devices of 12 and 17 mm were used for PDAs smaller than and larger than 2.5 mm, respectively, the ADO chosen was 2 mm larger than the smallest ductal diameter, and the NOc was selected using the manufacturer's chart. Ten minutes after implantation, follow-up aortic angiography was performed (Figures 1 and 2).
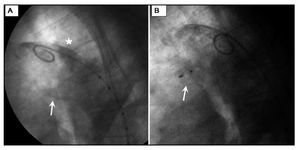
Figure 1. Ductal occlusion with Amplatzer Duct-Occluder. A: calcified patent ductus arteriosus (arrow) and calcified aortic wall (asterisk). B: Amplatzer Duct-Occluder (arrow).
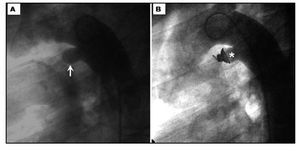
Figure 2. Ductal occlusion with Nit-Occlud coil. Patent ductus arteriosus (arrow) and Nit-Occlud coil (asterisk).
RESULTS
The PDA types observed in the study included type A (69.6%), type C (8.7%), type E (17.4%), and filiform (4.3%); 2 had wall calcification. The mean pulmonary artery pressure was 9-72 mmHg (mean, 15 mmHg), and the pulmonary and aortic artery diameters of the PDA were 1.8-5.8 (mean, 3.5) and 9-17 mm (mean, 15 mm), respectively. In the RAS group, occlusion was ruled out in 1 patient who had a mean pulmonary artery pressure of 72 mmHg with negative results in the balloon occlusion and 100% O2 test. The procedure was successfully completed in 7 of the 8 remaining cases, yielding an intention-to-treat success rate of 87.5%. In the patient with filiform PDA, the procedure failed because the diameter did not allow catheter advancement to implant a coil. In the ADO and NOc groups, implantation was successfully completed in all patients.
Immediate residual shunt was classified as a trace leak if the pulmonary end of the ductus showed slight contrast opacification, mild if the pulmonary artery but not the pulmonary valve was opacified, and moderate if the entire valve was delimited.4
In the RAS group, the immediate residual shunt was considered a trace leak in 2 cases and mild in 1; in the ADO group, mild in 2 cases and moderate in 1, and in the NOc group, mild in 1 case. The patients remained in a common ward and were discharged the day after the operation.
Table lists the occlusion rates following implantation of the devices. There were no cases of recanalization in any of the successfully occluded PDAs.5,6
Patients were followed up for 0.25 to 6.5 years (median, 2). The only immediate complication reported was inguinal bruising, and there were no late complications.
DISCUSSION
As in other series,7,8 our study population is small, in part because the condition is usually diagnosed and treated early in life. The most common PDA in our series was type A in the Krichenko9 classification, a finding consistent with reports by other authors.
Although PDA should be closed once diagnosed, the management of silent PDA is still controversial. Patients may be referred because PDA was observed on color Doppler ultrasound; however, the angiographic diameter is the determining factor for coil implantation, as in our patients.
The high percentage of immediate residual shunt and low incidence of postprocedure occlusion observed in the RAS group are similar to the findings reported by other authors.10,11 We stopped using this device in mid-1999 because other devices that have smaller sheaths and are simpler to implant became available, such as the ADO (AGA Medical Corporation, Golden Valley, Minnesota, United States) and the NOc (PFM, Cologne, Germany). We have no experience with the Sideris button device for ductal occlusion in adults because it is not available in our setting.12 Nor do we have experience with Gianturco coils because of the higher risk of migration and persistent residual shunt13 or the Amplatzer Muscular VSD occluder, which has been successfully used in patients with pulmonary hypertension.14
As has been reported in other studies,4,15 we also observed a high percentage of residual shunt in patients receiving the NOc, although the rate of postprocedure occlusion on immediate follow-up was substantially higher. We do not use this device in large PDAs (>4 mm) because of the greater probability of residual shunt or migration, although the manufacturer recommends it for PDAs with a diameter of up to 5 mm. In these cases, we prefer the ADO, which does not have these drawbacks.16
At the start of our experience (RAS group), 1 patient presented severe pulmonary hypertension with negative occlusion and hyperoxygenation tests; therefore, ductal closure was considered contraindicated. Nonetheless, current articles on the use of pulmonary vasodilator medication in patients with pulmonary hypertension and congenital heart disease report encouraging results.17-19
In our series, percutaneous closure of PDA in adults using several types of devices designed for this purpose, especially late-generation devices, was safe and effective, regardless of the morphologic and histologic characteristics of the defect.
Correspondence: Dr. R.E. Rios-Méndez.
Avda. Belgrano 1746, piso 5.o. (C1093AAS) Buenos Aires. Argentina.
E-mail: riosmendez@intramed.net.ar
Received October 30, 2008.
Accepted for publication July 15, 2009.