Atrial fibrillation (AF) continues to be the most common arrhythmia, with a prevalence of around 1% to 2% in the general population. In the setting of arrhythmia, it is the leading cause of morbidity and mortality, and the focus of the majority of scientific production. This year has seen the publication of information on the role of direct oral anticoagulants in the prevention of cardioembolic stroke in AF. There have been numerous efficacy and safety studies in clinical practice, most of which are multicenter retrospective studies, but which support the conclusions of previous clinical trials and reinforce the fundamental role of these drugs in the prevention of stroke vs vitamin K antagonists (VKA).1 In addition, the RE-CIRCUIT 2study demonstrated a lower rate of major bleeding when performing AF ablation without interrupting dabigatran therapy compared with conventional treatment with VKA. These findings confirm the safety of interventional cardiology procedures without the suspension of direct oral anticoagulants, as was already the case in clinical practice with VKA. Finally, direct oral anticoagulants have been compared with atrial appendage occlusion, showing that the death and stroke rates are very similar. Nevertheless, these results should be taken with caution and new studies are needed to confirm them.
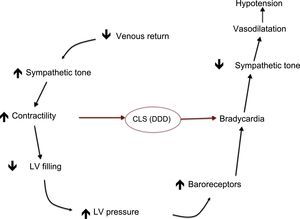
In the field of syncope, a Spanish group presented the SPAIN3 trial at the Congress of the American College of Cardiology. This trial evaluated the effectiveness of dual-chamber pacing with closed-loop stimulation algorithm for the treatment of vasovagal syncope in patients with recurrent episodes and a cardioinhibitory head-up tilt test (Figure 1). There was a significant reduction in syncope recurrence in patients with a pacemaker with this algorithm compared with patients with a pacemaker in DDI mode, confirming the results of previous studies. Even so, pacing therapy should not be generalized in vasovagal syncope, but reserved only for patients with highly recurrent episodes, significant clinical involvement, and a cardioinhibitory mechanism.
Closed loop stimulation diagram. The sensor detects the change in intracardiac impedance that occurs in the initial phase of vasovagal syndrome and activates sequential stimulation, which prevents bradycardia, decreased sympathetic tone, and hypotension. CLS, closed loop stimulation; LV, left ventricle.
In the field of inherited cardiomyopathies and sudden cardiac death (SCD), an outstanding study is that of the Brussels group on risk stratification of SCD in Brugada syndrome.4 In contrast to previous meta-analyses that only gave importance to syncope in the decision to implant automatic defibrillators in these patients, this group developed a risk scale in which not only the presence of previous syncope or cardiac arrest were significant, but also male sex, spontaneous Brugada pattern type 1 on electrocardiogram, sinus dysfunction, a direct family history of SCD, and inducible ventricular arrhythmias by programmed stimulation. These findings should be confirmed in larger multi-center studies, but will surely keep the debate on this difficult issue alive. In the area of channelopathies, a large randomized study demonstrated the efficacy of flecainide in reducing the arrhythmic burden of catecholaminergic polymorphic ventricular tachycardia.5 This study confirmed flecainide as a therapeutic alternative to beta-blockers, either in monotherapy or in combination, and as an attractive additional therapeutic tool in this disease in addition to beta blockers themselves, sympathectomy, and automatic defibrillator implantation. Finally, a study by a Spanish group was the first to identify mutations in the FLNC gene, which encodes filamin C, as a cause of arrhythmogenic myocardiopathy of the left ventricle.6 This relevant study advances our understanding of the etiopathogenesis of this entity and provides results that are applicable in the clinical setting, because it showed the families described had a high incidence of myocardial fibrosis, ventricular arrhythmias, and SCD. Filamin C is a cytoskeletal anchoring protein previously associated with hypertrophic cardiomyopathy and restrictive cardiomyopathy. However, this new study only included radical mutations, or truncations, establishing a strong phenotype-genotype correlation and confirming the prognostic value of genetic information in SCD risk stratification in these cases of left arrhythmogenic dysplasia.