Keywords
INTRODUCTION
Surgical treatment of multiple ventricular septal defects (VSDs) is complex, particularly with anterior and apical locations. Normally, several interventions are required, increasing the risk of adverse effects and death unrelated to the residual shunts. There is also a greater need for ventriculotomies and extensive sectioning to facilitate access. Percutaneous approach is also difficult, particularly in small children who have limited vascular access and may poorly tolerate the use of large devices. Percutaneous device placement under direct vision with extracorporeal circulation (ECC) and cardioplegia have not produced outstanding results.1 Amin et al2 reported perventricular closure with an Amplatzer device (AGA Medical Corporation, MN, United States of America) for VSD occlusion. The technique was updated by Bacha et al3 with excellent results in the first consecutive series of patients studied. The self-expandable double-disk device is made from nitinol mesh with the disks joined by a 7 mm waist. The diameter of the disks determines the size of the Amplatzer device.
CASE STUDY
A 15-month-old girl weighing 8 kg with multiple VSDs (large posterior muscular VSD and apical "Swiss cheese" septal defects) had received a pulmonary artery (PA) band during the neonatal period. One year later, definitive corrective surgery was planned. The echocardiogram revealed a posterior muscular defect and three other apical defects measuring 3 mm to 6 mm. These apical defects were the only defect on the left side of the septum. The PA band was correctly positioned, with a gradient of 80 mm Hg. Moderate right ventricular (RV) hypertrophy was apparent.
Informed consent was obtained before implanting the Amplatzer device.
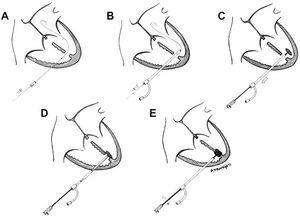
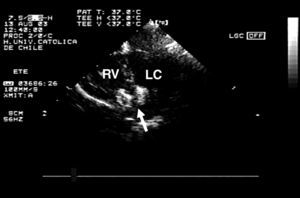
After medial sternotomy, the heart and great vessels were exposed. Under transesophageal echocardiographic (TEE) guidance with the heart beating, the RV free wall was punctured with an 18 G needle, avoiding the papillary muscles. An ultraflexible guidewire was introduced into the left ventricle (LV) through the largest apical defect (Figure 1A). An 8 F sheaf was then introduced into the LV over the guidewire, which was subsequently retracted (Figure 1B). The size of the device had to be 1 to 2 mm larger than the main VSD, thus an 8 mm Amplatzer device was chosen. After immersion in unheparinized blood for 20 minutes to allow the pores in the nitinol mesh to become blocked, the device was screwed onto the release system (cable). A 7 F sheaf was introduced under watertight seal, and advanced over the introducer. The sheaf was then tensed to allow deployment of the left disk (Figure 1C). The whole system was then tensed (cable and sheaf) so that the disk lay against the septum (Figure 1D). Then the other sheaf was withdrawn and the right disk was deployed. Monitoring with TEE showed that there was no residual shunting and that the disks were correctly positioned (Figure 2). The cable was then unscrewed and the device released (Figure 1E). The RV puncture site was repaired and we proceeded to conventional closure of the posterior muscular VSD with a synthetic patch. Finally, with ECC and aortic clamps in place, the AP band was withdrawn.
Figure 1. Schematic representation of perventricular closure of a ventricular septal defect with an Amplatzer device.
Figure 2. Four-chamber transesophageal echocardiogram showing that the device is appropriately positioned. Arrow indicates device attached to the release system; RV, right ventricle; LV, left ventricle.
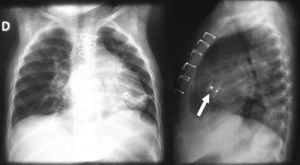
The patient recovered with no evidence of ventricular dysfunction or arrhythmias. Chest x-ray showed that the device and the disks were correctly deployed (Figure 3) and the echocardiogram before discharge from hospital showed no residual shunt. The girl was discharged after 5 days and remains well after 6 months of follow-up.
Figure 3. Postoperative chest x-ray. Arrow indicates device at apex of ventricular septum.
DISCUSSION
Despite progress in surgery, repair of certain types of VSD remains a challenge, even in hospitals with extensive experience.4,5
The technique we describe is reproducible and appears to be a promising alternative for patients with multiple VSDs, particularly with apical or anterior locations where access is difficult. It might also be an option in patients who have difficult vascular access but who are nevertheless candidates for percutaneous closure of VSD. Finally, it could be an interesting alternative for patients who require surgery for complex heart disease and who also have VSDs. This technique shortens ECC time and, more importantly, aortic clamping time, and so it represents an advance with respect to device placement techniques under direct vision. Despite the limited experience with this technique, the results are promising and it could become a treatment of choice.
Correspondence: Dr. P. Becker.
Marcoleta, 367 6o. Santiago de Chile. Chile.
E-mail: pbecker@med.puc.cl