Cryoballoon ablation (CBA) accounted for 19% of all ablation procedures for atrial fibrillation (AF) in Spain in 2012.1 Little information is available on the long-term outcomes of CBA.2 No data have been reported from low-volume centers, but such information is important because almost 80% of Spanish catheterization laboratories perform fewer than 50 AF ablations per year.1
We prospectively analyzed the outcomes of CBA as the first-line technique in a low-volume center. From November 2010 through June 2013, 63 patients were included (12 of whom were women; mean [standard deviation] age, 55 [10] years). Of these patients, 48 (76%) had recurrent paroxysmal AF, and 15 had short-lasting persistent AF (< 6 months). The mean left atrial diameter was 41 (4) mm, and 9 patients (14%) had structural heart disease; 11 patients (18%) had a score ≥ 2 on the CHA2DS2–VASc scale.
Oral anticoagulants were administered for at least 1 month prior to CBA and were continued for 1 month after the ablation procedure. After the first month, the decision to continue or stop anticoagulant therapy was based on the score on the CHA2DS2–VASc scale. Antiarrhythmic agents were continued for 3 months after the procedure (6 months in persistent AF).
Cryoballoon ablation was initially performed through double transseptal puncture, although single puncture techniques were used once the intraluminal circular catheter became available. The last 19 procedures were performed with the Advance Cryoballoon (Medtronic Inc.), which was the device that achieved the most homogeneous application to the whole circumference of the opening of the pulmonary vein. At least 2 applications were made per vein, with continuous phrenic pacing of the right pulmonary veins during ablation to monitor for the appearance of phrenic paralysis.
Procedure and fluoroscopy times were 168 (22) minutes and 30 (12) minutes, respectively. These times decreased significantly after the first 30 procedures (the procedure time was 189 [18] minutes for the first 30 procedures and 147 [34] minutes for subsequent procedures, while the fluoroscopy time was 35 [7] minutes for the first 30 procedures and 23 [5] minutes for subsequent procedures; P<.001). Electrical isolation was achieved in 224 of the 231 pulmonary veins identified (97%). The vessel most frequently left without isolation was the right inferior vein (4 procedures), in all cases because phrenic paralysis was detected in the superior right pulmonary vein during the application. Six patients had a common chamber (4 left, 2 right).
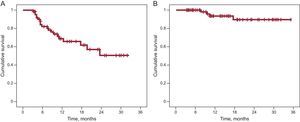
During the 12 (8) months of follow-up, recurrence of AF was reported in 20 patients (32%), with a rate of sustained sinus rhythm after 1 and 2 years of 69% and 51%, respectively. With antiarrhythmic agents, these rates increased to 94% and 86%, respectively (Figure). Symptomatic arrhythmic episodes persisted in 5 patients, AF was documented in 3 patients, and 2 patients had atrial flutter. All these patients underwent a further ablation procedure, this time with radiofrequency.
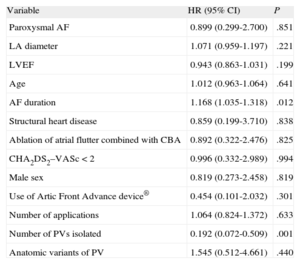
On multivariate analysis, the only independent predictor of recurrence of AF after CBA was time in AF prior to the procedure (hazard ratio [HR]=1.17; 95% confidence interval [CI], 1.035-1.32; P=.012). The number of pulmonary veins isolated was a protective factor (HR=0.19; 95% CI, 0.072-0.509; P=.001) (Table). Of the 4 patients without application to the right inferior pulmonary vein, 2 experienced recurrence.
Predictors of Arrhythmic Recurrence After Cryoballoon Ablation of Atrial Fibrillation
| Variable | HR (95% CI) | P |
| Paroxysmal AF | 0.899 (0.299-2.700) | .851 |
| LA diameter | 1.071 (0.959-1.197) | .221 |
| LVEF | 0.943 (0.863-1.031) | .199 |
| Age | 1.012 (0.963-1.064) | .641 |
| AF duration | 1.168 (1.035-1.318) | .012 |
| Structural heart disease | 0.859 (0.199-3.710) | .838 |
| Ablation of atrial flutter combined with CBA | 0.892 (0.322-2.476) | .825 |
| CHA2DS2–VASc<2 | 0.996 (0.332-2.989) | .994 |
| Male sex | 0.819 (0.273-2.458) | .819 |
| Use of Artic Front Advance device® | 0.454 (0.101-2.032) | .301 |
| Number of applications | 1.064 (0.824-1.372) | .633 |
| Number of PVs isolated | 0.192 (0.072-0.509) | .001 |
| Anatomic variants of PV | 1.545 (0.512-4.661) | .440 |
AF, atrial fibrillation; CBA, cryoballoon ablation; CI, confidence interval; HR, hazard ratio; LA, left atrial; LV, left ventricle; PV, pulmonary veins.
Statistical significance was set at P<.05.
Twelve complications were reported (19% of procedures). These were mainly phrenic paralysis in right pulmonary veins during the procedure (7 cases, 11%). These events were always transient, although resolution occurred late in 2 patients (9 and 12 months). After controlling the application times (240-180seconds) and temperature (limited to −55°C), no phrenic paralyses were reported in the last 28 ablations, with no impact on the efficacy of the procedure (P=.01). In addition, there was 1 transient ischemic attack due to air embolism, 1 asymptomatic thalamic stroke, and 2 minor episodes of hemoptysis. Finally, there was 1 case of atrioesophageal fistula, attributed to application (with the new cryoballoon) to the left inferior pulmonary vein. The patient died 1 month after the procedure.
The learning curve for CBA as the treatment of choice for recurrent paroxysmal AF or short-lasting persistent AF in small-volume centers is steep, as indicated by the high initial short-term efficacy and fast decrease in fluoroscopy and procedure times (P<.001) in our series. The rates of sustained sinus rhythm with and without antiarrhythmic drugs and the incidence of phrenic paralysis and severe complications (3 out of 63 cases [4.8%]) appear to be similar to those of other series with a larger number of patients.3–5 Although the incidence of complications may seem high, it was driven mainly by transient phrenic paralysis. This complication was eliminated when we limited the application time and minimum temperature reached (P=.01). Importantly, however, changes in the ablation parameters and the small number of patients in the series prevent affirmations about the steepness of the learning curve, particularly regarding the incidence of serious complications.