Hypercholesterolemia is a major risk factor for cardiovascular disease. Homozygous familial hypercholesterolemia (HoFH; OMIM #143890) is a rare disease caused by mutations in the low-density lipoprotein-cholesterol receptor gene. It is characterized by extremely elevated plasma low-density lipoprotein (LDL), tendon and skin xanthomas, and progressive atherosclerosis and results in death before 30 years of age if not treated.1 HoFH patients show varying cardiovascular disease risk and undoubtedly represent a particular cohort of cardiovascular disease patients. Accordingly, the standard algorithm for assessing cardiovascular risk, atherosclerotic burden, and inducible cardiac ischemia may not be suitable.1
Indeed, it is also clear that myocardial perfusion scintigraphy should be prescribed with caution to limit the lifetime radiation exposure. On the other hand, cardiac magnetic resonance stress testing is not widely available or well-standardized.
Based on these considerations and on previous work,2 we decided to perform the myocardial ischemia follow-up of 4 HoFH siblings by using dual-imaging stress echocardiography to measure the wall motion and Doppler-derived coronary flow reserve (CFR) of the mid-distal left anterior descending artery (LAD) during dipyridamole stress echocardiography (DiSE-CFR).
CFR can be measured by several invasive and noninvasive techniques, quantitatively by positron emission tomography or by Doppler flow wire during coronary angiography or as coronary blood flow velocity during DiSE. Of these techniques, transthoracic echocardiography allows the highly accurate recording of flow velocity in the mid-distal LAD. CFR in the LAD during DiSE has excellent concordance with invasive Doppler flow wire readings and good reproducibility.3 For this reason, it is now more commonly used in the stress echo laboratory; the intra- and interobserver variabilities for Doppler recording measurements are <10%.4
CFR has been proven to exhibit powerful and additive prognostic value in large-scale multicenter studies involving patients with known or suspected coronary artery disease.3 A CFR reduction can indicate significant epicardial coronary artery stenosis (typically when the CFR is <1.8) or coronary microvascular disease2,4 and therefore probably constitutes a further prognostic index.3,4
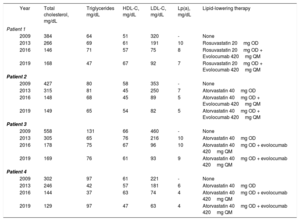
In the present study, we examined 4 HoFH siblings who were compound heterozygotes for LDL receptor gene mutations5: 1 woman (patient 1), 2 twin sisters (patients 2 and 3), and 1 man (patient 4), who were 26, 24, and 18 years old, respectively, on admission to our lipid clinic in 2009. They all had xanthomas in the Achilles tendon since adolescence that was overlooked until they were 19 years old. They had been treated, at best, with dietary approaches and multidrug therapy until 2013 when, upon the addition of evolocumab to high-intensity statin therapy, a satisfactory lipid profile was obtained (table 1).
Lipid profile and lipid-lowering therapy in 4 siblings who are compound heterozygotes for LDL receptor gene mutations
| Year | Total cholesterol, mg/dL | Triglycerides mg/dL | HDL-C, mg/dL | LDL-C, mg/dL | Lp(a), mg/dL | Lipid-lowering therapy | |
|---|---|---|---|---|---|---|---|
| Patient 1 | |||||||
| 2009 | 384 | 64 | 51 | 320 | - | None | |
| 2013 | 266 | 69 | 61 | 191 | 10 | Rosuvastatin 20mg OD | |
| 2016 | 146 | 71 | 57 | 75 | 8 | Rosuvastatin 20mg OD + Evolocumab 420mg QM | |
| 2019 | 168 | 47 | 67 | 92 | 7 | Rosuvastatin 20mg OD + Evolocumab 420mg QM | |
| Patient 2 | |||||||
| 2009 | 427 | 80 | 58 | 353 | - | None | |
| 2013 | 315 | 81 | 45 | 250 | 7 | Atorvastatin 40mg OD | |
| 2016 | 148 | 68 | 45 | 89 | 5 | Atorvastatin 40mg OD + Evolocumab 420mg QM | |
| 2019 | 149 | 65 | 54 | 82 | 5 | Atorvastatin 40mg OD + Evolocumab 420mg QM | |
| Patient 3 | |||||||
| 2009 | 558 | 131 | 66 | 460 | - | None | |
| 2013 | 305 | 65 | 76 | 216 | 10 | Atorvastatin 40mg OD | |
| 2016 | 178 | 75 | 67 | 96 | 10 | Atorvastatin 40mg OD + evolocumab 420mg QM | |
| 2019 | 169 | 76 | 61 | 93 | 9 | Atorvastatin 40mg OD + evolocumab 420mg QM | |
| Patient 4 | |||||||
| 2009 | 302 | 97 | 61 | 221 | - | None | |
| 2013 | 246 | 42 | 57 | 181 | 6 | Atorvastatin 40mg OD | |
| 2016 | 144 | 37 | 63 | 74 | 4 | Atorvastatin 40mg OD + evolocumab 420mg QM | |
| 2019 | 129 | 97 | 47 | 63 | 4 | Atorvastatin 40mg OD + evolocumab 420mg QM | |
HDL-C, high-density lipoprotein-cholesterol; LDL, low-density lipoprotein; LDL-C, low-density lipoprotein-cholesterol; Lp(a), lipoprotein(a); OD, once a day; QM, every 4 weeks.
All patients were asymptomatic for angina and/or dyspnea. However, they showed slight alterations in ventricular repolarization (slight ascending ST-segment in DII-DIII-aVF and from V4 to V6 rapidly regressing during the recovery phase) in ergometric stress testing while being negative for inducible myocardial ischemia. We thus followed up these patients with serial DiSE-CFR to evaluate both myocardial ischemia and CFR in the LAD.
At the first DiSE, patient 4 was positive for myocardial ischemia (left ventricular hypokinesia of the apical segments and of the middle anterior wall) and for CFR alterations (absence of detectable coronary blood flow in the LAD under rest conditions). Subsequent coronary angiography revealed the presence of chronic occlusion of the anterior descending artery and> 70% stenosis in the first diagonal branch with a Rentrop 1 collateral circle; these lesions were treated with percutaneous coronary revascularization (percutaneous transluminal coronary angioplasty/stenting of the LAD artery and of the first diagonal branch). DiSE-CFR performed at 10 months follow-up failed to detect inducible ischemia and the CFR was “normal” (2.22).
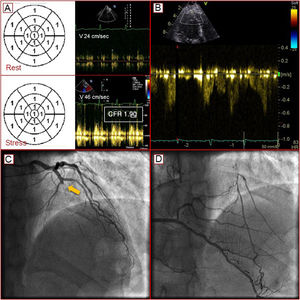
At the first DiSE-CFR, patient 1 showed no inducible ischemia, based on wall motion criteria, but a slight CFR reduction (1.90) compatible with coronary microvascular disease. The addition of evolocumab to statin therapy achieved a satisfactory lipid profile (table 1). Tests able to identify the presence of high-risk plaque6 were delayed because the patient was asymptomatic for angina and dyspnea. At 2 years follow-up, the same test showed a LAD blood flow with increased velocity and inverted direction, suggesting a collateral coronary circulation. Subsequent coronary angiography revealed the presence of chronic occlusion of the LAD in the presence of valid (Rentrop 3) coronary collateral flow (figure 1). The patient was treated with percutaneous coronary revascularization (percutaneous transluminal coronary angioplasty/stenting of the LAD).
A: normal wall motion in the presence of a slight reduction in coronary flow reserve (CFR) compatible with coronary microvascular disease. B: CFR in the left anterior descending artery with an increased speed and inverted direction due to the presence of collateral coronary circulation. C: chronic occlusion of the anterior descending artery (arrow). D: intracoronary collateral flow.
The 2 female twins (patients 2 and 3) showed no abnormal values in the DiSE-CFR tests.
The feasibility of stress echocardiography is undermined by its poor acoustic window and the specific contraindications to dipyridamole (presence of severe conduction disturbances, asthma, and a resting systolic blood pressure <100 mmHg). Furthermore, we specifically measured the CFR of the LAD. The 3-coronary artery approach would undoubtedly be more fruitful but it remains too technically challenging. In addition, we must also consider the lifetime radiation exposure. However, the combination of wall motion and CFR into a single test with transthoracic echocardiography enables the simultaneous imaging of LAD flow and regional wall motion.
In the clinical setting for HoFH, stress echocardiography wall motion criteria and CFR may be considered a valid tool to assess inducible myocardial ischemia, LAD stenosis, and coronary microvascular dysfunction.4
The patients described here underline the need for the individualized follow-up of HoFH patients aimed at updating the risk of acute cardiovascular disease events and balancing the test cost:benefit ratio with very valid concerns about lifetime radiation exposure.
The authors are grateful to Mascia Pianelli and Roberta Luciani for their excellent work in the apheresis unit.