Implantable cardioverter-defibrillator (ICD) implantation is the first-line therapy for patients with Brugada syndrome (BrS) in secondary prevention after malignant ventricular arrhythmias (VA) or sudden death. After ICD implantation, the incidence of VA and high-energy shocks is high,1 thereby resulting in a significant impact on quality of life and prognosis.
Recently, there has been increasing evidence of the use of quinidine as an adjuvant treatment2 in ICD patients with frequent episodes of malignant VA. Quinidine inhibits outward transient K current (Ito1), which is believed to result in homogenization of the repolarization of the epicardial layers by restoring the action potential dome where Ito1 currents are predominant, as in the epicardium of the right ventricular outflow tract.3 Despite quinidine treatment, some patients continue to experience episodes of VA. The presence of monomorphic ventricular tachycardias (MVT) in BrS patients has been previously described, is linked to variable mechanisms and is sometimes responsive to antitachycardia pacing (ATP).4 There have been no reports of the modification of malignant VA to MVT amenable to ATP termination in patients under quinidine treatment. We present a series of 3 patients showing a possibly novel effect of quinidine on the pattern of VA in patients with BrS.
We reviewed all episodes of VA among 29 patients with BrS under treatment with quinidine due to frequent ICD shocks included in a Spanish national registry.5 We describe 3 patients from this registry who showed MVT terminated by ATP (Table). The general characteristics of the registry have been described elsewhere.5
Patient Characteristics
| Patient | Age, y | Sex | Familial BrS or SD | Atrial fibrillation | Clinical presentation | ECG pattern | RBBB | EPS | Genetic testing | Reason for quinidine initiation | Time ICD-quinidine (mo) | Follow-up on quinidine (mo) | Drug | Adverse effects |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 24 | M | No | No | SD | Spontaneous type 1 | No | Not performed | Not performed | Multiple syncopes for NS PVT | 19 | 12 | Hydroxiquinidine 1000 mg | Yes, diarrhea |
| 2 | 64 | M | No | No | Multiple syncopes | Drug induced type 1 | No | VF inducible | BrS and ARVC negative | Multiple shocks (4) for PVT | 24 | 50 | Hidroxiquinidine 1000 mg | No |
| 3 | 41 | M | No | No | SD | Spontaneous type 1 | Yes | Not performed | BrS negative | Multiples shocks (9) for VF | 48 | 72 | Quinidine bisulphate 600 mg | No |
ARVC, arrhythmogenic right ventricular dysplasia; BrS, Brugada syndrome; ECG, electrocardiogram, EPS, electrophysiologic study; ICD, implantable cardioverter-defibrillator; M, male; NS PVT, nonsustained polymorphic ventricular tachycardia; PVT, polymorphic ventricular tachycardia; RBBB, right bundle branch block; SD, sudden death; VF, ventricular fibrillation.
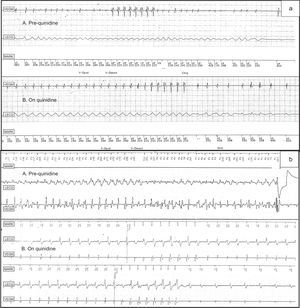
Patient 1 presented with sudden death at the age of 24 years. He had spontaneous type 1 Brugada pattern in leads V1-V2. An ICD was implanted. After 19 months, he experienced an episode of syncope due to polymorphic ventricular tachycardia (PVT) and near-syncope episodes due to unsustained PVTs. Hydroxiquinidine (1000mg/d) was started. The patient had not experienced any syncope or PVT since quinidine initiation, but after 12 months of follow-up he had 2 episodes of MVT terminated with a burst of ATP (Figure A).
A: Pre-quinidine: syncopal polymorphic VT (230ms cycle length) degenerating into VF treated with unsuccessful ATP and spontaneously terminated; On Quinidine: monomorphic fast VT accelerating from initial 320ms to 290ms successfully terminated by ATP. B: Pre-quinidine: polymorphic VT treated with shock; On Quinidine: monomorphic slightly irregular slow VT (390ms cycle length) terminated by first ATP ramp after 3 failed ATP bursts (last failed bursts is shown before the successful ATP ramp. ATP, antitachycardia pacing; Chrg, charge; Dvrt, diverted therapy; LECG, “leadless” ECG; MARK, channel mark; V-Detect, ventricular detection; VEGM, ventricular electrogram; V-Epsd, ventricular episode; VF, ventricular fibrillation; VP, ventricular pacing; VS, ventricular sensing; VT, ventricular tachycardia.
Patient 2 had experienced several episodes of syncope at the age of 64 years. He showed type 2 Brugada pattern (type 1 after ajmaline). An ICD was implanted. Following ICD implantation, he had 4 episodes of ventricular fibrillation (VF) treated with shocks and therefore hydroxiquinidine was started (1000mg/d). During 50 months of follow-up, he had no VF episodes; nevertheless, he had 62 episodes of MVT that were all terminated with bursts of ATP.
Patient 3 had an episode of aborted sudden death at the age of 41 years; his baseline electrocardiogram showed type 1 Brugada pattern. Four years after ICD implantation, quinidine bisulphate (600mg) was initiated due to 9 episodes of VF treated with shocks. After 72 months of follow-up and no VF episodes, he had 1 episode of MVT (cycle 280ms) terminated with a burst of ATP (Figure B). At present, the 3 patients are still on quinidine treatment (a mean treatment duration of 45 months) without no episodes of PVT or VF.
The most relevant finding of this case series is the modification of VA pattern in these BrS patients treated with quinidine, with no recurrence of PVT/VF but instead with MVT terminated by ATP without requiring high-energy shocks.
These results introduce new data on the analysis of the mechanism of VA in patients with BrS and the interaction between these mechanisms and the effect of quinidine.
According to the most accepted theories, the net outward shift of ionic currents during the end of phase 1 of the action potential results in an accentuation of the action potential notch, leading to the dispersion of repolarization and creating the substrate for phase 2 reentry and VA.3 Recently, it has been suggested that alterations of depolarization are the mechanisms underlying the clinical manifestations of BrS: late potentials and fractionated electrogram have been observed in the epicardium of the right ventricular outflow tract.6 Structural abnormalities have been described such as hypertrophy, fibrosis, and fatty infiltration in the right ventricular outflow tract, related to slowed conduction.
The presence of subtle structural abnormalities could favor the development of reentrant circuits and explain the finding of MVT in these patients. In this setting, quinidine could stabilize right ventricular outflow tract excitability and facilitate the conversion of malignant VA into more stable and regular MVT responsive to ATP. The positive response to ATP reinforces the idea of a reentrant circuit with excitable gap; it might be that quinidine-mediated slowing of conduction could facilitate the ATP burst to propagate to the circuit, depolarize the excitable gap, and extinguish reentry.
In view of this finding, it can be proposed that patients with BrS treated with quinidine due to frequent ICD shocks might benefit from the introduction of a fast VT zone with 1 or 2 ATP bursts since it has proven effectiveness in MVT termination and shock reduction. Further research is needed to confirm this uncommon VA pattern in BrS after quinidine treatment and elucidate its mechanism.