Ischemic heart disease is the leading cause of heart failure (HF).1 Ventricular aneurysms are more common in persons with a prior myocardial infarction, resulting in progressive ventricular remodeling and HF.1
Our aim is to describe 2 new techniques for ventricular restoration using the Parachute® percutaneous device (CardioKinetix; Menlo Park, United States) and the PliCath HF® epicardial device (BioVentrix; San Ramon, United States) in patients with HF and a previous ventricular aneurysm secondary to extensive myocardial infarction. We also discuss the clinical outcomes of 4 patients treated with this new technology.
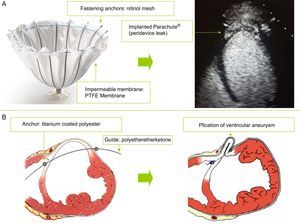
The Parachute® percutaneous device (Figure A) has a self-expanding nitinol structure with anchor-shaped filaments at its distal end, which allow apposition to the ventricular wall and anchoring in the endomyocardium. The metal structure is covered by an impermeable polytetrafluoroethylene membrane, enabling occlusion of the aneurysm cavity, excluding the ventricular aneurysm.2,3 A large gauge introducer (16 Fr) is inserted via an arteriofemoral access point, facilitating advancement of the device's delivery system. For the retrograde arterial approach, the left ventricular apex is reached via the aortic valve. Once optimal positioning is achieved, the release and apposition of the device against the ventricular wall is completed by inflating a balloon.
The PliCath HF® epicardial device (Figure B) is implanted using median sternotomy and fluoroscopic guidance. The right ventricle is accessed by inserting a specially-designed curved needle through the anterior ventricular wall and crossing the left ventricular cavity and the septum. To insert this needle, a guide composed of a high-strength biocompatible material (polyetheretherketone) is advanced to the pulmonary artery, and is then used to insert a catheter. A fastening anchor is introduced through this catheter and is then retracted until it comes into contact with the right ventricular face of the septum. The catheter is then removed and another fixing anchor is placed on the outer face of the ventricular wall via the point of needle insertion. Several pairs of anchors are implanted in a similar way until optimal plication of the ventricular aneurysm is achieved. The anchors are composed of a highly resistant material (titanium) and are coated with polyester to facilitate endothelialization.
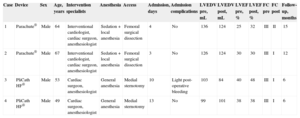
The 4 patients had extensive anterior myocardial infarction and signs and symptoms of HF. All underwent imaging tests, showing ventricular aneurysms and lack of viability in the left anterior ventricular wall. The first 2 were successfully treated using the Parachute® device, and the others using the PliCath® device. The baseline characteristics of the procedure and follow-up are summarized in the Table.
Baseline Characteristics of the Intervention and Follow-up
| Case | Device | Sex | Age, years | Intervention specialists | Anesthesia | Access | Admission, days | Admission complications | LVEDV pre, mL | LVEDV post, mL | LVEF pre, % | LVEF post, % | FC pre | FC post | Follow-up, months |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Parachute® | Male | 64 | Interventional cardiologist, cardiac surgeon, anesthesiologist | Sedation + local anesthesia | Femoral surgical dissection | 4 | No | 136 | 124 | 25 | 32 | III | II | 15 |
| 2 | Parachute® | Male | 67 | Interventional cardiologist, cardiac surgeon, anesthesiologist | Sedation + local anesthesia | Femoral surgical dissection | 3 | No | 126 | 124 | 30 | 30 | III | I | 12 |
| 3 | PliCath HF® | Male | 53 | Cardiac surgeon, anesthesiologist | General anesthesia | Medial sternotomy | 10 | Light post-operative bleeding | 103 | 84 | 40 | 48 | III | I | 6 |
| 4 | PliCath HF® | Male | 49 | Cardiac surgeon, anesthesiologist | General anesthesia | Medial sternotomy | 13 | No | 99 | 101 | 38 | 38 | III | I | 6 |
FC, New York Heart Association functional class; LVEF, left ventricular ejection fraction; LVEDV, left ventricular end-diastolic volume.
The anteroapical region is particularly vulnerable to swelling due to its greater curvature and thinner structure. Expansion and thinning of this region, as in the case of ventricular aneurysm following extensive myocardial infarction, increases the ventricular radius and thus wall tension. This eventually degenerates into inefficient ventricular contractility and finally HF.3,4
Classical surgical ventricular restoration procedures using ventricular aneurysm resection were developed to reduce volume and obtain an elliptical ventricular shape, thus halting progressive ventricular remodeling. However, these procedures had a high complications rate related to cannulation, cardiopulmonary bypass, and left ventriculotomy. They were therefore abandoned in routine clinical practice because of their high surgical risk.4
New approaches have recently emerged for ventricular restoration, which are less invasive and provide the benefits of restoration therapy without exposing the patient to the greater risk of classical surgical techniques.
The PARACHUTE IV study2,3 (NCT00573560, United States and NCT01286116, Europe) evaluated the Parachute® device. Thirty-nine patients with ventricular aneurysms, HF (functional class II-IV), and left ventricular ejection fraction between 15% and 40% were recruited in the US and Europe. The procedure was attempted in 34 patients and was successful in 31. In patients who were successfully treated, diastolic volume was significantly reduced, and 85% of them showed improved functional class. Of the 31 patients, 2 (6.5%) died of cardiac causes in the first 6 months, and no other cardiac deaths occurred during the 3-year follow-up.
CONFIGURE-HF (NCT01568138) is a safety study that is evaluating the PliCath HF® device. This is a multicenter, multinational safety study in patients with previous ventricular aneurysms, ventricular dilation (indexed telesystolic volume ≥ 60mL / m2) and a left ventricular ejection fraction of 15% to 40%. The primary objective of this study is to assess the overall rate of serious adverse events related to the device.
These initial studies will report the feasibility and safety of these new techniques. We are still at an early stage in the development of this technology, but if further clinical trials confirm that if these techniques are safe and able to halt ventricular remodeling, they may be a therapeutic alternative for these patients.