Keywords
INTRODUCTION
One of the most alarming complications of surgical or interventional treatment for aortic coarctation or angioplasty of the pulmonary arteries is the development of an aneurysm. We describe 2 such cases occurring in our unit and the application of a novel therapeutic alternative for treatment.
CASE STUDIES
Case 1
An 18-year-old patient had undergone surgery for aortic coarctation (AoCo) in the neonatal period with the Waldhausen technique (aortoplasty with left subclavian artery autograft) and had progressed favorably. At the age of 5 he presented signs of recoarctation, moderate mitral stenosis, and mild aortic stenosis, and was successfully treated at that time by balloon angioplasty. In a follow-up study at age 15, a 1-cm aneurysm was found at the distal end of the aortic isthmus with mild recoarctation. Magnetic resonance (MR) monitoring at one year showed enlargement of the aneurysm, and the patient was referred for surgery. The procedure consisted in simple closure of the aneurysm mouth with protected sutures.
During follow-up at age 18, MR again showed considerable recoarctation, for which cardiac catheterization was indicated. Angiography showed a 7.2-mm aneurysm, an area consistent with a small dissection and moderate aortic recoarctation, with a 40-mm Hg gradient, 8-mm diameter of the coarctation segment, 13.6-mm aortic isthmus, and 20-mm descending aorta.
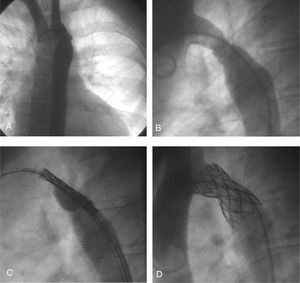
As an alternative to a third surgical procedure, covered stent implantation was proposed for combined treatment of both lesions. The following technique was performed: a 0.035-inch super-stiff Amplatz "J" guidewire was inserted using a percutaneous femoral artery approach and positioned in the ascending aorta, together with a 65-cm 14 French Mullins sheath. Through the sheath we implanted a 34-mm long expanded polytetrafluoroethylene (PTFE)-covered stent (covered CP stent) premounted on a BIB® balloon with an 18-mm diameter outer balloon and a 9-mm inner balloon. The stent was reinforced by manual crimping and the balloon-stent combination was protected with a plastic tube, in order to go through the hemostatic valve of the Mullins sheath and impede posterior displacement of the stent. With the aid of manual angiography through the sheath, the stent was positioned without problems. The balloon was inflated manually, first the inner and then the outer balloon. Angiographic monitoring showed exclusion of the aneurysm and slight narrowing of the stent, with no hemodynamic repercussions. The angiographic and pressure data are shown in Table 1 and Figure 1. At 48 h the patient was discharged with a small residual gradient and no complications.
Figure 1. A and B: aneurysm and recoarctation. C: stent positioned in the area of coarctation of the aorta and the aneurysm (inflation of the inner balloon) D: stent now in place; exclusion of the aneurysm and increased diameter of the coarcted segment are evident.
Case 2
A patient with tetralogy of Fallot underwent corrective surgery at age 2, consisting in widening of the right ventricular outflow tract with a transannular GoreTex® patch (ventriculotomy extended to the origin of the left pulmonary artery [LPA]) and closure of the interventricular communication with a Dacron patch. At long-term follow-up, progressive stenosis was observed at the origin of both pulmonary arteries, predominantly in the LPA. When the patient was 10 years old and asymptomatic, a trans-stenotic gradient of 60-mm Hg was detected on echocardiography. Therapeutic catheterization and stent implantation was decided. The patient weighed 43 kg and body surface area was 1.26 m2.
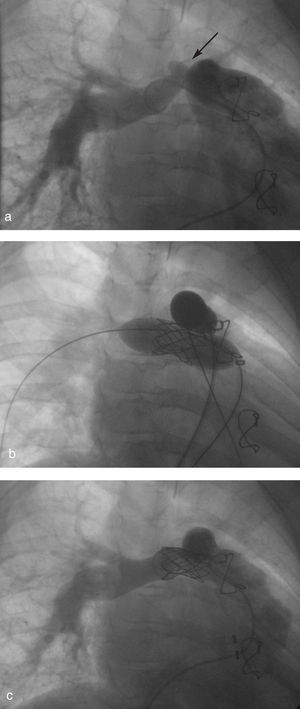
Angiographic study of both pulmonary arteries revealed double stenosis at their origin and an aneurysmal sac in the superior wall of the right pulmonary artery, measuring 7 mm at the base and 4 mm in height. Covered stent implantation was decided. Angiographic images of the implantation of both stents are shown in Figure 2.
Figure 2. Angiography showing the following: a) aneurysm in the right pulmonary artery (arrow); b) simultaneous implantation of the covered stent and the ITI stent; and c) posterior angiography with exclusion of the aneurysm and increased diameter of the pulmonary arteries.
The technique used was as follows: 2 Mullins sheaths (12 and 11 Fr) were positioned in the right and left femoral veins. Through the 12-Fr sheath, a 28-mm long, GoreTex®-covered CP Stent (Numed®), mounted on a 6x4-mm BIB balloon, was positioned in the right pulmonary artery (RPA) at the site of the stenosis to cover the aneurysm. Because of slight proximal displacement of the stent over the balloon (caused by resistance and the angle of the sheath as it passed through the right ventricular outflow tract), the stent had to be repositioned before inflation. To achieve this, the proximal edge of the stent was made to coincide with the tip of the Mullins sheath, and, while firmly holding the balloon-stent combination, the sheath was pushed until the stent was centered over the radio-opaque marks on the balloon. A 26-mm ITI MEGA (EV3®) stent mounted on a 16x3-mm BIB balloon was inserted in the LPA, positioning it at the site of the stenosis. Once proper positioning of both stents had been confirmed by manual angiography through the sheath, the stents were manually inflated simultaneously.
As described in Table 2, a clear decrease in right ventricular pressure as well as a significant increase in the diameters of both pulmonary arteries was achieved, with exclusion of the aneurysm on selective pulmonary angiography.
Currently, one year after the procedure, the patient is enjoying a normal life style with echocardiographic follow-up assessments showing similar right and left ventricular pressures, and with no evidence of an aneurysm.
REVIEW AND DISCUSSION
The anatomical and histological criteria upon which the development of an aneurysm is based are inferred from microscopic observation of the surgically resected segments of aortic coarctation. There is a severe decrease and disarray of the elastic tissue in the vessel wall, and in two-thirds of cases, a histological alteration known as "cystic medial necrosis" of the vessel wall. This factor results in weakening of the aorta wall and favors the formation of an aneurysm following injury to the vessel during angioplasty or surgery.1 This histological lesion is very similar to that described for idiopathic dilation of the aorta in patients with Marfan syndrome,1,2 although this finding has not been mentioned by other authors.
Injury to the intima and media of the vessel is produced during angioplasty, and this should lead to a high incidence of aneurysms. Nevertheless, the incidence of this complication varies. In an angiographic study following balloon dilation, Rao et al3 saw a process of remodeling of the aortic contour, which in some way could be a determining factor in the formation of aneurysms.
Incidence
The reported incidence of aneurysms is quite high, reaching 43% to 50% in earlier series.4,1 Parks et al4 found 20 aneurysms in 39 patients operated between 1976 and 1987 (50%). Experience with the technique has probably had some influence on this, since the incidence after both surgery and angioplasty is lower in more recent series, with rates as low as 2%-10%. Fletcher et al5 reported 2 cases among 102 patients (1.9%) and Mendelsohn et al6 found 3 cases among 59 patients receiving angioplasty for native aortic coarctation (5%). Suárez de Lezo et al7 obtained an incidence of 10% in coarctations treated with surgery or angioplasty. It can be considered a limiting factor of the study that long-term follow-up with angiography was not systematic in the patients with angioplasty. Rao et al,3 however, performed angiographic follow-up in a group of patients and found a 5% incidence (3 aneurysms in 58 patients). The frequency of aneurysm development in patients treated with stents for AoCo is even lower.8,9
Some authors3,10 believe that the decrease in this high incidence is related to the emphasis on extreme care with the balloon dilation technique, avoiding excessive distension of the coarcted segment. Current recommendations for the choice of balloon advise that the diameter should not exceed the diameters of the aortic isthmus or the diaphragmatic aorta, and not exceed three-fold the diameter of the coarcted segment.11 In addition, extremely careful manipulation of the catheter and guidewire in the recently dilated region decreases the risk of injury to the treated segment, and consequently, the potential for aneurysm formation. Lastly, the appearance of new noninvasive imaging techniques, such as MR, has contributed to the follow-up of these patients.
In this regard, the criteria establishing which MR features indicate an aneurysm have had to be defined. Most of the related studies consider an aneurysm to be present when there is a vessel wall deformity with a diameter 1.5 times the diameter of the diaphragmatic aorta.1 Hence, MR imaging can be considered a suitable, non-invasive technique for the detection of aneurysms and its use is recommended for follow-up of all patients with this condition, whether treated surgically or by therapeutic catheterization.1,11
In a study conducted among adults,12 an attempt was made to investigate prognostic factors for aneurysm formation following coarctation surgery. According to the results, one-third of aneurysms occur in the ascending aorta and therefore would have no relation with the surgery. In addition, a clear association was found with the presence of a bicuspid aortic valve, a common occurrence in this disease.
A Cheatham-platinum (CP) stent covered with PTFE membrane was used in our patients. The CP stent is manufactured with an alloy of 90% platinum and 10% of iridium. Theoretically, this combination is more malleable and has adequate radial strength, which is enhanced by the design in a "zig" pattern. The CP stent has rounded edges, decreasing the risk of balloon rupture or injury to the vessel wall; in addition, the platinum component makes it more radio-opaque.13 The stent is covered with PTFE, which is secured with cyanoacrylate adhesive at 4 points on the outside of the stent on each edge. In one of our cases the CP Stent was premounted on a BIB balloon. The utility of the CP covered stent resides in the characteristics of its wall. Since it is covered, it occludes the entry orifice in the vessel wall and excludes the aneurysm.
The literature contains descriptions of similar cases in which the covered stent resulted in successful outcomes,14-16 a fact that encourages us to present it as a good alternative treatment option. The recent report of a fatal case17 involving a 65-year-old patient with an aneurysm and coarctation has prompted some authors to recommend the covered stent as the first choice11,18,19 in native coarctation and in older patients with a higher risk of aneurysm formation or aortic rupture.
Some limitations of the covered stent must also be mentioned: a) the patient's age, since its use is limited in small children; b) the vascular access, because of the need to use a large sheath (13, 14, or 16 Fr); c) the risk of excluding emerging collateral vessels, such as the subclavian, spinal or others in the case of the abdominal aorta; and d) it is not certain whether future redilation of a covered stent will be effective. These factors require caution when using these procedures, which should only be performed in interventional cardiology units with considerable experience and the means to resolve potential complications.
Correspondence: Dr. R. Bermúdez-Cañete.
Servicio de Cardiología Pediátrica. Hospital Ramón y Cajal.
Carretera de Colmenar Viejo, km 9,100. 28034 Madrid. España.
E-mail: r_bermudez@terra.es