The increasing use of ventricular assist devices as a bridge to heart transplantation improves survival in candidates for transplantation. Intravenous unfractionated heparin (UFH) is routinely used to prevent the thromboembolic complications associated with the pump and support cannulae. Occasionally, UFH can trigger a specific immunological response, characterized by the development of antibodies against UFH/platelet factor 4 complex, activating platelets, and leading to heparin-induced thrombocytopenia (HIT). In such cases, UFH must be switched to an alternative anticoagulation, usually parenteral direct thrombin inhibitors.1
After 3 months, anti-UFH/platelet factor 4 antibodies are usually undetectable, and the risk of recurrent HIT on re-exposure to heparin is reduced. For patients with acute or subacute HIT requiring cardiopulmonary bypass (CPB) surgery, clinical guidelines recommend delaying surgery, when possible, until the disappearance of antibodies, and then using UFH again. If surgery cannot be delayed, a different anticoagulant can be used (grade 2C).1 However, patients with HIT who have short-term ventricular assist devices cannot wait for the antibodies to disappear. Therefore, the treatment of these patients presents a clinical challenge. In the case of CPB, early re-exposure to UFH can make HIT worse. However, there is limited experience with the use of direct thrombin inhibitors during CPB, and a significant increase in hemorrhage has been described, due to the complexity of intra-operative monitoring of the anticoagulant effect and the lack of antidotes.2
We describe our experience with 4 patients who required a ventricular assist device (Levitronix Centrimag) due to acute heart failure and who developed HIT and underwent transplantation using UFH within a few weeks.
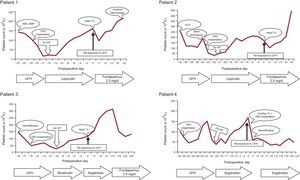
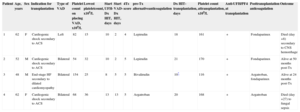
Patient characteristics and trends in platelet counts are summarized in the Table and in the Figure, respectively. A more detailed description of each case is provided in the supplementary material.
Summary of Patient Characteristics
| Patient | Age, years | Sex | Indication for transplantation | Type of VAD | Platelet count on placing VAD, x109/L | Lowest plateletcount, x109/L | Start UFH-Dx HIT, days | Start VAD-Dx HIT, days | 4Ts score | pre-Tx alternativeanticoagulation | Dx HIT-transplantation, days | Platelet count attransplantation, x109/L | Anti-UFH/PF4 at transplantation | Posttransplantation anticoagulation | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 62 | F | Cardiogenic shock secondary to ACS | Left | 82 | 15 | 10 | 2 | 4 | Lepirudin | 18 | 161 | + | Fondaparinux | Died (day +8) secondary to CNS hemorrhage |
| 2 | 52 | M | Cardiogenic shock secondary to ACS | Bilateral | 54 | 32 | 10 | 2 | 5 | Lepirudin | 21 | 170 | + | Fondaparinux | Alive at 50 months post-Tx |
| 3 | 48 | M | End-stage HF secondary to dilated cardiomyopathy | Bilateral | 154 | 25 | 8 | 5 | 5 | Bivalirudin | 10* | 116 | + | Argatroban, fondaparinux | Alive at 24 months post-Tx |
| 4 | 62 | F | Cardiogenic shock secondary to ACS | Bilateral | 68 | 36 | 13 | 13 | 5 | Argatroban | 20 | 168 | + | Argatroban | Died (day +27) to fungal sepsis |
ACS, acute coronary syndrome; CNS, central nervous system; Dx HIT, heparin-induced thrombocytopenia; F, female; HF, heart failure; M, male; Tx, transplantation; VAD, ventricular assist device.
According to the 4Ts score,1 all patients had at least an intermediate pretest clinical probability of HIT. All patients had positive determination of anti-UFH/PF4 antibodies on particle gel immunoassay (PaGIA, DiaMed-AG, Switzerland). Unfortunately, the results of platelet function tests were not available. However, the diagnosis of HIT was supported by a progressive recovery of baseline platelet values in all patients after the switch from UFH to direct thombin inhibitors (despite the persistence of other possible causes of thrombocytopenia). Heart transplantation (cardiorenal transplantation in 1 patient) was done between 10 and 20 days after the diagnosis of HIT. At the time of transplantation, all patients had a platelet concentration similar to baseline, although determination of anti-UFH/PF4 antibodies remained positive. None of the patients had thrombotic complications associated with HIT before transplantation. In all patients, the alternative anticoagulant therapy (lepirudin, bivalirudin, or argatroban) was stopped 2hours before surgery, and UFH alone was used during the CPB period, as is standard practice. Postoperatively, after adequate hemostasis was ensured, the patients were restarted on alternative anticoagulation therapy with fondaparinux or argatroban. No patients showed signs of recurrent HIT. Two patients died. One patient died on day 8 posttransplantation (spontaneous intracerebral hemorrhage) and 1 died on day 27 (fungal sepsis). The other 2 patients are alive at 50 and 24 months after transplantation.
This series is one of the largest studies in patients with ventricular assist devices as a bridge to transplantation and confirms reports of other patients with acute HIT requiring early CPB and UFH.3,4 It would be ideal to have access to platelet function tests, using washed platelets, allowing at least confirmation of platelet normalization before transplantation (even if anti-UFH/PF4 antibodies remain detectable). However, these techniques are very complex and are not available in most centers-even in tertiary centers-in Spain.5 On this subject, a recent systematic review concluded that the presence of anti-UFH/PF4 antibodies in patients who underwent cardiac surgery was not predictive of thromboembolic complications or postoperative death.6
In patients with ventricular assist devices who develop HIT without associated thrombosis, our experience shows that, when platelet function testing is unavailable, early re-exposure to UFH alone during cardiac transplantation and CPB could be an alternative to the use of direct thrombin inhibitors, provided that the preoperative platelet count has recovered and that an alternative anticoagulant treatment is restarted postoperatively.