Keywords
INTRODUCTION
Some forms of congenital heart disease require systemic pulmonary shunts with an expanded polytetrafluoroethylene tube, also known as a modified Blalock-Taussig (mBT) shunt, to ensure patients survive until corrective or palliative surgery for the underlying illness is possible. This shunt is usually clamped during later surgical interventions although they may be left permeable for various reasons.1-3 Percutaneous occlusion has been performed electively using different techniques employing various types of coils and devices.2-8
The Amplatzer Vascular Plug (AVP) is an occlusive device that, although originally designed to embolize peripheral arteriovenous vascular malformations,9 has been used in other situations.9-13
We report our experience of using the AVP to occlude mBT shunts.
METHODS
We obtained previous, informed written consent. All interventions were performed under general anesthetic, using right femoral artery approach, antibiotic prophylaxis and administering 100 U/kg sodium heparin in children and 5000 U the adults. We implanted one AVP in each patient.
Patient 1
A girl with D-transposition of major vessels, left ventricular hypoplasia, sizable atrial septal defect, small ventricular septal defect, and infundibular pulmonary stenosis, who had undergone left subclavian-pulmonary anastomosis (LSCPA) that became dysfunctional and, hence, motivated right systemic-pulmonary anastomosis (RSPA). During Glenn bidirectional surgery, the RSPA was clamped but the contralateral anastomosis was not because the approach was too difficult. Before Fontan type surgery, embolization of the LSCPA was indicated.
Patient 2
A boy with pulmonary atresia and intact septum who underwent RSPA and, later, surgical valvulotomy. In the follow-up, we confirmed adequate development of the right ventricle enabling him to maintain biventricular physiology; severe right pulmonary artery stenosis related with the site of the mBT suture; and permeable RSPA.
Patient 3
A man with a single ventricle with a double entrance, sizable atrial septal defect, severe pulmonary stenosis, vena cava inferior draining into right atrium, absence of right superior cava vein, innominate and left superior cava veins draining into coronary sinus. He had undergone RSPA between the innominate artery and right pulmonary artery and later underwent Glenn bidirectional surgery (anastomosis of left superior cava vein with left pulmonary artery) and clamping of the pulmonary artery trunk. In the follow-up, we confirmed the development of a large fistula between the innominate and right superior pulmonary veins that was occluded with an Amplatzer Duct-Occluder. Before Fontan surgery, we attempted to occlude the RSPA with 2 Gianturco coils that migrated towards the left pulmonary artery a few minutes after implantation, leading to their extraction with a snare catheter.
Procedure
We located the end of the mBT with a 5 Fr right coronary Judkins catheter through which we advanced a soft-tipped metal guidewire to attain a position inside the anastomosis. Over the metal guidewire we changed the catheter for a 5 Fr right coronary guidewire in the first 2 patients and a 6 Fr guidewire in the third, positioning the end inside the mBT; through the catheter guidewire we advanced and implanted the device (Table). After 10 min, we conducted aortography or selective mBT angiography to verify occlusion (Figure 1). In patient 2, we also performed balloon angioplasty of the right pulmonary artery with a venous approach.
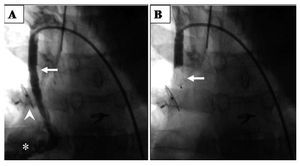
Figure 1. Occlusion of RSPA with AVP (patient 3). A: permeable RSPA (arrow), right pulmonary artery (asterisk); ADO occluding fistula between innominate and right superior pulmonary veins (arrowhead). B: AVP (arrow) occluding RSPA. ADO indicates Amplatzer Duct-Occluder; AVP, Amplatzer Vascular Plug; RSPA, right systemic-pulmonary anastomosis.
RESULTS
Angiographic occlusion of mBT was confirmed in all patients. Fluoroscopy times were 10, 11, and 9 min respectively. The first 2 patients were discharged at 24 h and the third underwent next-day Fontan type surgery. No complications arose during the procedure.
Three and a half years later, patient 1 underwent repeat catheterization due to suspected fenestrated total cavopulmonary shunt dysfunction, which showed occluded LSCPA, permeable subclavian and left pulmonary arteries and AVP with the same configuration and location as at device implantation (Figure 2). In patient 3, an aortic tear occurred during sternotomy and was sutured; at 3 days post-surgery he presented acute hemodynamic depression due to hemorrhaging from the site of the aortic lesion, causing death.
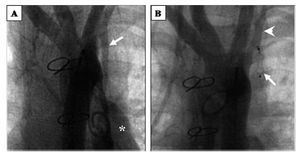
Figure 2.LSCPA occluded with AVP (patient 1). A: permeable LSCPA (arrow), left pulmonary artery (asterisk). B: aortography, 3.5 years later; LSCPA occluded with AVP (arrow) conserving "dog's bone" configuration and permeable left subclavian artery (arrowhead). AVP indicates Amplatzer Vascular Plug; LSCPA, left subclavian-pulmonary anastomosis.
During follow-up, no recanalization of the mBT occurred, as was corroborated by color spectral Doppler ultrasound.
DISCUSSION
While mBT is usually clamped during Glenn or Fontan surgery, the option of maintaining permeability is sometimes preferred. This may be the case in high-risk patients, such as those with very small caliber pulmonary arteries, in whom it is decided to leave a pulsatile bidirectional Glenn shunt so as to induce pulmonary artery growth.1 Another motive, as in patient 1, may be the existence of technical or anatomic difficulties of access2 (eg, peri-mBT fibrosis).
A variety of materials have been used to embolize mBT.2,3,6-8,14 Both the Duct-Occlud and the Nit-Occlud coil (PFM, Cologne, Germany)15,16 have been used to embolize mBT and large collateral aortopulmonary arteries, respectively, but we consider that the length, relative rigidity and lack of internal constricture of mBT limit its optimal reconfiguration once deployed. In our third patient, 2 Gianturco coils migrated on first attempt to clamp the RSPA, which leads us to agree with other authors who believe the greatest disadvantage of these coils is the significant incidence of migration.6,14
Although the Amplatzer Duct-Occluder has been used successfully in these cases,2,5 it is considered less useful in tube-like structures, as it was specifically designed and configured for type A ductus arteriosus.10
The AVP is a relatively new cylindrical occluder,17 with characteristics typical of Amplatzer® devices (AGA Medical Corp.) except that it has a more dense mesh and does not contain thrombogenic material. The AVP is deployed through small caliber catheters, making it particularly advantageous in the arterial approach in small patients like two in our series. Following the manufacturer's recommendation, in all the patients we implanted devices of a diameter 50%-75% greater than the caliber of the luminal conduit. Given its morphology, the AVP permitted a greater, more homogeneous support surface within the prosthesis.
We coincide with other groups in considering percutaneous mBT embolization with AVP a safe, effective procedure that may avoid surgery or, in the context of corrective or palliative surgery, reduces morbidity and surgery time. However, given the limited number of communications on this type of patient,3-5,9 further follow-ups and more experience are needed before defining indication for AVP to occlude prosthetic vascular structures.
Correspondence: Dr R.E. Rios-Méndez.
Avda. Belgrano 1746, 5.o (C1093AAS) Buenos Aires. Argentina.
E-mail: rrmendez@yahoo.com.ar
Received August 17, 2008.
Accepted for publication January 7, 2009.