Keywords
CASE REPORT
After 3 days of bed-rest for influenza-like symptoms, a 68 year-old diabetic man who smoked and suffered high blood pressure presented at our emergency department with angina, dyspnea, and palpitations upon slight exertion. His blood pressure was 105/85 mm Hg and his heart rate 110 bpm. Tachypnea without rales was evident and a mild, tricuspid regurgitating murmur was detected. An electrocardiogram revealed left bundle branch block. A chest x-ray was normal. Analysis revealed hypoxemia (pO2, 50 mm Hg), hypocapnea (pCO2, 31 mm Hg), hyposaturation (90%), increased D-dimers (3.1 ng/mL), and troponin T levels (0.4 ng/mL). Computed tomography (with contrast) confirmed a diagnosis of pulmonary embolism (PE) via the disclosure of clots at the bifurcation of the common pulmonary artery and in its main branches (Figure 1). An echocardiogram revealed a dilated right ventricle (RV) associated with dyskinesia of the medial segment of the free wall (Figure 2). Pulmonary blood pressure was 55 mm Hg. The source of the embolism was a right femoral venous thrombosis (remains of clots were found by echo-Doppler analysis).
Fig. 1. Helicoidal thoracic computed tomography with contrast (angio-CT). Axial view of the common pulmonary artery and branches. Clots can be seen at the bifurcation of the common pulmonary artery (curved black arrow) and in its main branches (straight black arrow).
Fig. 2. Transthoracic echocardiogram. Apical view of the 4 heart chambers. A paradoxical systolic protrusion (dyskinesia) of the free wall of the right ventricle can be appreciated (black arrows). RA indicates right atrium; LA, left atrium; RV, right ventricle; LV, left ventricle.
Treatment involved peripheral thrombolysis with plasminogen tissue activator. An echocardiogram at 72 h showed an improvement in the RV segment anomalies and pulmonary blood pressure (35 mm Hg). After favorable progression the patient was discharged and treatment with acenocoumarol and bisoprolol prescribed.
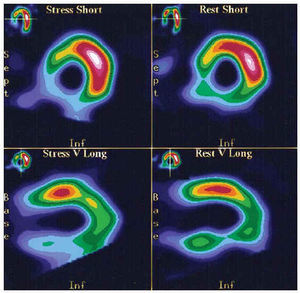
One month later the patient complained of angina upon strenuous exertion. An echocardiogram was normal. Myocardial scintigraphy (with dypiridamole) revealed an extensive infero-posterior perfusion defect (Figure 3). After 6 months of anti-coagulation therapy, coronary angiography revealed an occlusion of the proximal segment of the right coronary artery with abundant collateral circulation. The patient was treated with aspirin, statins, and beta-blockers. No complications were evident at one year of follow-up.
Fig. 3. Myocardial perfusion scintigraphy with 201Tl under pharmacological stress with dypiridamole. Axial views of the short axis and sagittal views of the long axis. An extensive perfusion defect compatible with severe infero-posterior ischemia can be appreciated.
DISCUSSION
Acute, massive PE (20%) is associated with serious hypoxemia and hemodynamic instability. Mortality can be high if no treatment is provided (up to 30%). The initial progress of the disease appears to be better with thrombolysis than with heparin alone; it is therefore generally recommended in such cases.1-4
The majority of patients with PE are stable at the time of diagnosis, although this does not necessarily imply a benign outcome: the prognosis varies depending on whether the RV is affected, as determined by echocardiography. Normotensive patients with no echographic anomalies (60%) have a practically zero likelihood of developing serious complications. However, those who are initially stable but who show RV dysfunction are at greater risk of clinical deterioration and recurrence of embolism; mortality among these patients is 5%. In the absence of large clinical trials, some authors have suggested that thrombolysis might also be beneficial in patients with subclinical RV dysfunction (i.e., it might lead to an improvement in RV function and a possible reduction in morbidity and mortality). Thrombolytic treatment should, therefore, be provided on an individual basis (i.e., as long as there is no increased risk of hemorrhage and in the absence of cardiorespiratory disease with pulmonary hypertension).1-5
As well as being helpful in making a differential diagnosis, an echocardiogram can provide important information directly related to the PE.2-6 In addition to the classical findings (dilation and hypokinesia of the RV with leftward septal protrusion, dilation of the pulmonary arteries, dilation of the inferior vena cava [without collapse], tricuspid regurgitation velocity >3 m/s, RV ejection acceleration time <90 ms), a pattern of regional RV dysfunction has been described in acute PE. This is more specific in terms of diagnosis, and consists of hypokinesia or akinesia of the medial segments of the free wall (in apical views of the 4 heart chambers); apical mobility is preserved.7-9 The mechanism proposed for explaining these anomalies include the drag of the hyperdynamic left ventricle on the right ventricular apex, the spherical morphology adopted by the RV in order to equilibrate the tension of the wall in front of an acute increase in afterload, and the ischemia provoked by the sudden increase in parietal stress.
In acute thromboembolic cor pulmonale, the combination of a reduction in coronary perfusion (through the reduction in cardiac output and aortic pressure) and the increased right ventricular afterload leads to myocardial ischemia. This can aggravate RV dysfunction and perpetuate hemodynamic instability. Clear signs of myocardial ischemia have been described in PE: increased cardiac troponin levels, RV perfusion defects in scintigraphy with 201Tl, and even isolated infarctions of the RV in necropsies of patients with massive embolisms but no coronary artery disease.10,11
In the present case, dysfunction of the RV affected the free wall but in the form of dyskinesia, a more noticeable segmentary anomaly than those previously described. In addition to the mechanisms mentioned, this dysfunction can be explained by the coexistence of a coronary artery lesion compromising the perfusion of the RV. It is concluded that RV dysfunction in patients with acute thromboembolic cor pulmonale might be aggravated by underlying coronary artery disease.
Correspondence: Dr. A. Rubio Alcaide.
Urbanización Montesol, 53. 29730 Rincón de la Victoria. Málaga. España.
E-mail: alvaro13@terra.com