Keywords
INTRODUCTION
Familial hypercholesterolemia (FH) is a hereditary problem that affects 1 in 400-500 people in the general population. It is caused by mutations in the gene coding for the receptor for low density lipoprotein and is transmitted in an autosomal dominant fashion. At least 50% of the first degree relatives of an affected person inherit the problem.1 The importance of its diagnosis lies in the high incidence of premature cardiovascular disease2,3 and the increased risk of cardiovascular death4,5 with which it is associated, especially in people under 40 years of age.4 It is estimated that if appropriate treatment is not provided, at least 50% of men and 20% of women with FH will suffer a coronary episode before 50 years of age.2-4 There is now sufficient evidence to show that cardiovascular mortality and morbidity can be reduced by the use of statins in high risk patients with cardiovascular disease,6-8 although for patients with FH such evidence is scarce.9,10 However, it is probable that early diagnosis and adequate lipid lowering treatment could reduce these problems, especially in people under 60 years of age9; they are therefore highly recommendable with respect to this population.11 Nonetheless, the high cost of statins and the lifetime treatment required render such primary prevention controversial from an economic point of view.
No studies have been published on the cost-effectiveness of lipid-lowering treatment in patients with FH. The aim of the present work was to determine the cost-effectiveness of different therapeutic strategies for the prevention of cardiovascular disease in patients with FH, using a model comparing atorvastatin alone, or in combination with ezetimibe, with normal clinical practice (CP).
METHODS
Design
This study involved a longitudinal populational cost-effectiveness model with life expectancy as the time horizon. The study subjects were a cohort of patients diagnosed with FH showing the same profile as recorded in the Spanish FH Registry.2 A preventive lifetime lipid-lowering intervention for the reduction of cardiovascular risk, composed of 5 alternative treatments, was analyzed from the viewpoint of the Spanish health system.
Patients
The patient cohort was simulated from the data for patients included in the Spanish FH Registry2 with a genetic diagnosis of FH and for whom sociodemographic, physiological, lifestyle, cardiovascular background, comorbidity, and treatment information was complete. The final sample included 881 patients, 44% of whom were men. The mean age of patients was 48 (18-82) years.
Intervention
Patients received 1 of the following lipid-lowering treatments for the reduction of cardiovascular risk:a) CP as detailed in the Registry (Table 1 shows the different statins used and their clinical doses according to normal clinical practice for the years covered by the Registry [1999-2002]); b) atorvastatin 40 mg alone (A40);c) atorvastatin 40 mg plus ezetimibe 10 mg (A40+E10);d) atorvastatin 80 mg alone (A80); or e) atorvastatin 80 mg plus ezetimibe 10 mg (A80+E10).
Model
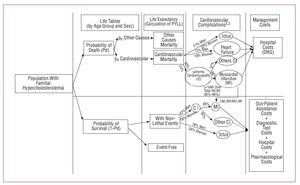
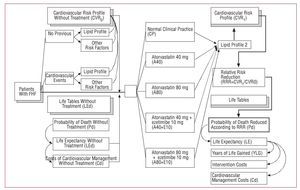
Actuarial methodology was used to calculate the potential years of life lost (PYLL) due to cardiovascular disease with respect to cardiovascular risk. For each patient the annual costs of treatment were imputed, as well as the costs corresponding to the consumption of resources used in the management of cardiovascular complications. Figure 1 shows the rationale of the model; Figure 2 describes the model in detail.
Figure 1. Rationale of the model.
Figure 2. Structure of the cost-effectiveness model.
Effectiveness
Using life tables for the Spanish population, life expectancy was calculated taking into account the sex and age group of all patients in the sample as though they belonged to the general population.12,13 In the determination of the PYLL due to FH it was assumed that: a) the increase in all-cause mortality was due to increased cardiovascular mortality, and that the distribution of deaths of other cause was no different to that seen in the general population; b) the genetic defect associated with FH is expressed over the lifetime of the patient; therefore, the different distribution of mortality with respect to the general population is the same in carriers of both sex and for each age group; c) the different all-cause mortality of the FH patients compared to the general population is obtainable by assuming a standardized mortality rate (SMR) per age group and sex of 1.59 (95% confidence interval [CI], 1.07-2.26)5; and d) once the all-cause and cardiovascular mortality of the general population is known,13 plus the SMR for the FH population, the different mortality of the FH patients can be calculated, along with their cardiovascular mortality, their life expectancy, and PYLL without treatment, compared to the general population.14 Table 2 shows the results of these calculations.
The effect of treatment was estimated by the reduction from the initial cardiovascular risk (CVR0) resulting from the reduction in low density lipoprotein cholesterol (LDL-C). The cardiovascular risk of each patient was calculated using the Framingham Heart Study15 equations for primary and further events. The LDL-C-lowering effect of the therapeutic statins was obtained from efficacy data in the literature.16 The cardiovascular risk modified by treatment (CVR1) allowed the estimation of the reduction in relative risk in the form of a coefficient (CVR1/CVR0) corresponding to a reduced probability of cardiovascular death over the next 5 years. Since the treatments followed were lifetime treatments, and since the probability of cardiovascular death was reduced in each of the 5 year periods until the end of life expectancy, the probability of all-cause death must also fall, thus modifying life expectancy. It was also assumed that treatment with statins until the end of life has a constant effect independent of age, and that no patients abandoned treatment.
The majority of the patients in the Registry had been prescribed a lipid-lowering treatment. The modified life expectancy of each was therefore calculated on the basis of the therapeutic efficacy of the treatment received according to data in the literature.16 The difference between the PYLL with and without intervention in each subject represents the number of years of life gained (YLG) with CP. This process was repeated to calculate the YLG associated with each treatment option in the intervention. In order to express the results as present-day values, a discount rate of 3% per year on the PYLL17 was assumed.
COSTS
Total costs were deemed to be the cost of the intervention plus the costs of managing FH and potential cardiovascular events. The intervention costs were obtained as the sum of the annual costs of treatment until the end of life expectancy. The annual cost per patient was €573.31 for A40, €1117.63 for A40+E10, €573.12 for A80, and €1117.44 for A80+E10. The costs of CP per patient were calculated from the treatment costs for the established doses over 1 year as shown in the Registry, always assuming the use of the most cost-effective commercial products.
The costs of managing FH were calculated assuming a consumption of assistance resources (medical and hospital assistance), diagnostic tests, and the pharmacological management of cardiovascular events. The resources used per patient/year depended on the incidence of cardiovascular events, the type of such events (myocardial infarction, other ischemic heart disease, ictus, heart failure), and their lethality.
Cardiovascular complications were simulated from the epidemiological profile of cardiovascular diseases for the Spanish population18,19; each patient was attributed, for his/her remaining years of life, the consumption of resources corresponding to the sum of those necessary to manage each fraction contemplated in this profile, weighted by populational prevalence. Figure 1 describes the annual fractions considered and their weight in the cost of management.
The information used to establish the resources consumed in the management of events was obtained by consultation with experts of different specialties. It was assumed that lethal events consumed hospital resources alone. Non-lethal events were understood to also consume extra-hospital resources. The hospital resources were obtained from combined clinical management groups, diagnosis-related groups (DRG). The hospital management costs of a complication were calculated as the sum of the DRG costs weighted by frequency20 (Table 3). Table 4 shows the resources and medical costs taken into account.21-24 Table 5 shows the pharmacological resources used, estimated from information supplied by experts and the calculation of the annual consumption necessary for each group of medications according to the prescriptions made and their price (extracted from different databases).25,26 Table 6 shows the total annual costs of managing FH and cardiovascular events.
All prices were adjusted to their costs in Spain in 2005. A 6% annual discount on these costs was taken into account.27
Sensitivity
Scenario analyses were performed with respect to SMR3: a) baseline scenario: central estimate for SMR and mean value of costs; b) best case scenario: upper limit of SMR and management costs 10% lower; and c) worst case scenario: lower limit of SMR and management costs 10% higher. A sensitivity analysis was also performed in which the discount rate for both costs and results was 6%.
RESULTS
Table 7 shows the results for costs and effectiveness.
Baseline Scenario
Assuming an SMR in FH of 1.59 with respect to the general population, the PYLL expected are 7551 (8.6 [interval, 5.4-9.4])/patient).
Treating with CP compared to no treatment led to an improvement (after the annual discount of 3%) of 1.97 (0-4.75) YLG. The cost of intervention (after the annual discount of 6%) was €5321 (0-41350), and of management €23 389 (13 686-54 505); the latter made up 81.5% of the total €28 710 (14 206-56 183).
With A40, the YLG compared to no treatment was 2.59 years (1.19-4.05). The cost of intervention per patient was €8237 (2560-9899) and of management €22 333 (13 412-49 459), 4.5% less than with CP. The total cost was €30 569 (23 311-53 931).
Treatment with A40+E10 was associated with a YLG/patient of 3.38 (1.67-5.16) with respect to no treatment. The cost of intervention per treatment was €16 057 (4990-19 297) and of management €20 047 (13 026-44 420), 14.3% less than with CP. The total cost was €36 104 (25 837-54 703).
Treatment with A80 was associated with a YLG/patient of 2.75 (1.29-4.29), an intervention cost of €8234 (2559-9896), and a management cost of €21 899 (13 339-48 572), 6.4% less than with CP. The total cost was €30 133 (23 235-53 044).
Treatment with A80+E10 was associated with a YLG/patient of 3.62 (1.82-5.48) with respect to no treatment. The intervention cost was €16 054 per patient (4990-19 294) and the management cost €19 262 (12 891-42 453), 17.6% less than with CP. The total cost per patient with this treatment was €35 317 (24 477-53 778).
Table 8 shows the results for effectiveness, costs, and efficiency for the different treatments and CP.
Treatment with A40 was associated with a YLG/patient of 0.62 for an additional cost of €1859/patient with respect to CP, an incremental cost-effectiveness (ICE) of €3012/YLG. Treating with A40+E10 added 1.41 YLG/patient and cost €7394 more than CP, resulting in an ICE of €5250/YLG. With A80 a mean 0.78 YLG/patient were gained over CP at an additional cost of €1423, resulting in an ICE of €1821/YLG. Finally, with A80+E10 the gain was 1.64 YLG/patient, at a cost of €6607 more than with CP, resulting in an ICE of €4021/YLG.
Sensitivity Analysis
The most unfavorable scenario was that when general and cardiovascular mortalities were similar to that of the general population (SMR=1.07) with cardiovascular management costs 10% lower; this minimized benefits and places this type of preventive strategy in doubt. Although the YLG are reduced to nearly half, the differential costs increase and the ICE is at least doubled compared to the baseline scenario; to obtain 1 YLG costs €7941 euros with A40, €12 221 with A40+E10, €5699 with A80, and €9978 with A80+E10.
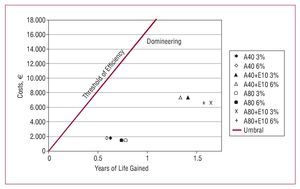
In the most favorable scenario, in which patients with FH die at a rate 2.26 times that of the general population, the preventive strategies improved the gain in YLG over CP, reducing differential costs to around those of CP in some cases (€352 in the case of A80). The resulting ICE was €1241 for A40, €2589 for A40+E10, €319 for A80, and €1616 euros for A80+E10. In the analysis in which the discount rate for costs and results was the same at 6%, the results were not substantially changed. Figure 3 shows the outcome for the baseline scenario when applying different discount rates to the results.
Figure 3. Baseline scenario: cost-effectiveness values with an annual discount rate of 3% and 6% for YLG, and of 6% for costs.
DISCUSSION
To our knowledge, this is the first study involving patients with FH designed to determine the cost-effectiveness of a high dose statin (atorvastatin) either alone or in combination with other drugs. Substituting normal CP for patients with FH by a therapeutic strategy based on atorvastatin alone or in combination with ezetimibe would lead to a gain in health, although this would depend on the initial status of the patient and the previous treatment received. The gradation of the results, with higher values for the combined therapies and higher doses of atorvastatin, and the agreement between the different sensitivity analyses, show that any of the higher dose treatments are valid.
Compared with CP, the total costs of the treatments examined were always higher. This is because CP can involve patients receiving no preventive treatment or lower doses, and because survival (extra YLG) leads to further treatment costs. The costs of managing FH and cardiovascular events are inversely proportional to the effectiveness of treatment since this reduces the probability of these events (compared to CP, A80 offers the best results).
The ICE expresses the extra cost of obtaining 1 YLG more than with CP. According to their ICE values, the monotherapies and higher doses of atorvastatin appear to be the most efficient. Any of the treatments in any scenario is cost-effective in comparison with CP according to the threshold of efficiency usually taken into account -$20 000/YLG (€16 848/YLG at December 12, 2005)28-30; preventive treatment is therefore justified in all patients with a genetic diagnosis of FH. For those patients at highest cardiovascular risk, treatment should be adjusted to their needs and complemented with other therapies.
The model used is based on the different cardiovascular mortality shown by patients with FH, which conditions an SMR different to that of the general population. The reference model used (which came from a Dutch report)5 is a new approach to the study of different mortality due to the effects of FH that, although it might be questioned in terms of its methodology and precision, provides an estimate with intervals of variability. The present model assumes that this different mortality may also be the case in other geographical areas with a similar population structure in which risk factors (mutations) are present. In addition, uncertainty is determined via a sensitivity analysis, approximating the mortality of patients with FH to that of the general population (the worst case scenario).
There is no evidence regarding how to extrapolate treatment effects to the end of life expectancy. This means that indirect data have to be used and several assumptions made. Given the different SMR compared to the general population, life tables allow estimates to be made regarding life expectancy, as well as the calculation of the PYLL owing to cardiovascular disease. After their calculation, the most plausible model is that which permits the effects of the different interventions on PYLL due to cardiovascular disease to be assessed.2
The presumption that a lifetime preventive treatment is worthwhile in genetic diseases manifested throughout life is logical, and is commonly made in economic models of statin use in cardiovascular disease.28-31 Associated with more reservations is the presumption that the effect of treatment is maintained over life and that no-one abandons treatment or has it changed. The present model is static and simulates the hypothetical case that initial conditions persist during the expected lifetime, thus projecting the current health status forward. It is not a dynamic model that varies depending on interaction variables or intermediate results; the assumptions required would go far beyond the evidence available. Rather, its objective is to provide information regarding the differences that would result from using different treatment alternatives. Therefore, owing to the parsimony of the model, adverse events were not modeled. Idiosyncratic adverse events would have an equivalent effect on all arms of the model since CP treatment usually includes the use of statins (Table 1). Adverse events dependent on dosage would have a greater effect in the arms involving A80, approximating their results to those of the A40 arms under the efficiency threshold. In any event, the incidence of adverse events is reduced: 2.3% of patients experience an increase in liver enzyme levels (not related to inherent liver problems), 5 out of 100 000 person per year develop myopathy, and 1.6 in every 100 000 persons per year develop rhabdomyelosis.32
Approximating the cardiovascular risk via the use of the Framingham equations is an approach that has been used before in studies on FH.33 The present model does not use the original equations but rather later versions that estimate the primary and secondary risk of an event. These were used for 2 reasons: a) a diagnosis of FH is very commonly established after an event at a young age; and b) there is a greater risk of a cardiovascular event among those patients who have already suffered one. In addition, people with FH are at high cardiovascular risk since they are born.4 Therefore, it would be incorrect to estimate the risk of an event with formulae that apply to the general population (such as the REGICOR equation adapted for the Spanish population, or the SCORE equation). Even so, using these equations with age groups outside of those with which these equations were developed (30-55 years) can cause a bias in the results. They can be used with patients aged under 30 years since studies exist that show the relationship between hypercholesterolemia and ischemic cardiomyopathy in people under this age.33 More questionable is their use with older people since the latter relationship is attenuated in the elderly.33 In addition, there is no evidence of any effect of treatment for older people. However, the present study uses the Framingham equations in an indirect manner, controlling other risk factors, in order to transform the proportional reduction in LDL-C into a relative reduction of cardiovascular risk and to be able to recalculate the PYLL. This methodology is applied equally in each arm of the intervention; there can therefore be no bias with respect to any particular intervention.
The first assumption was related to the study population: the cost per patient results from the imputation of the mean cost weighted by the prevalence of cardiovascular events, and the costs of the management of cardiovascular complications until the end of life expectancy. This assumption was made taking into account that the cost per patient is a valid central estimate of the situations that can arise with respect to the consumption of resources for the management of cardiovascular disease. The second assumption referred to the prevalence of lethal and non-lethal cardiovascular events, for which published values referring to the epidemiology of cardiovascular disease in Spain were used.18,19 Their use implies that the only modifier with respect to time is the different probability of death as determined by age and sex since the proportional distribution of cardiovascular events is constant. The variation in incidence of cardiovascular events with respect to age and sex is therefore modulated. Their use also implies an equivalent proportional reduction in the incidence of any cardiovascular event due to treatment. At the end of the day, the limitation associated with these implications affects the consumption of resources and costs, but not effectiveness. This limitation was tested in the sensitivity analysis with scenarios in which the cost of management was 10% higher or lower than the central value.
Finally, the number and type of resources consumed per cardiovascular event was estimated using the criteria of experts. This, as well as being limited in terms of scientific evidence, implies the absence of variability in clinical practice and a smaller spread of costs. In addition, it implies a constant form of management over time for each type of event—which is rather improbable. The exploratory and informative nature of the model used, plus the fact that the assumptions made must translate into costs, justify these assumptions being made. The variability in clinical practice translated into management costs may be considered robust according to the sensitivity analysis. Finally, the consideration of the mean cost incorporates populational elements that confer a representative value on the cost for the Spanish national health system. This value can be extrapolated to other years and has a certain stability despite changes in management resources.
CONCLUSIONS
The results of this cost-effectiveness analysis show that the use of protocols based on atorvastatin at doses of 40 and 80 mg in patients with FH is associated with a positive YLG result at a socially acceptable cost in comparison with normally accepted international thresholds. The greatest cost-effectiveness is achieved with atorvastatin 80 mg monotherapy. The addition of ezetimibe to treatment with atorvastatin 80 mg can provide additional positive effects for an acceptable increase in costs.
ABBREVIATIONS
CP: clinical practice
FH: familial hypercholesterolemia
ICE: incremental cost-effectiveness
PYLL: potential years of life lost
SMR: standardized mortality rate
YLG: years of life gained
Correspondence:
Dr. J. Fernández de Bobadilla.
Avda. de Europa, 20B, 28108 Alcobendas. Madrid. España.
E-mail: jaime.fernandez@pfizer.com
Received July 31, 2007.
Accepted for publication December 11, 2007.