Physicians practicing in the modern era are confronted with an ever aging and comorbid population. A working knowledge of the principles of frailty and its application to distinct medical conditions can help the clinician tailor recommendations to optimize both patient-centered goals and traditional outcomes. In this article, we will define frailty and synthesize the recent evidence on its role in affecting outcomes across the spectrum of cardiovascular disease (CVD).
Frailty is defined as a state of susceptibility in which a person has decreased physical reserve that leads to a greater likelihood of an adverse outcome when a stressor is applied. The concept of frailty came to prominence after a seminal publication by Fried et al, in which frailty was described as low overall physical activity level, unintentional weight loss, slow walking speed, fatigue, and loss of physical strength.1,2 Later works broadened this definition to include a variety of other factors, ie, comorbid medical conditions, loss of independence for activities of daily living, low albumin levels, and cognitive impairments.3,4
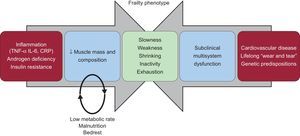
The pathophysiologic basis of frailty involves an interplay of aging-associated biologic changes and long-term “wear and tear” leading to subclinical organ dysfunction.5,6 The application of a stressor (in the form of an illness) makes “subclinical” disease become “clinical” and may result serious adverse outcomes (Figure).7
Two of the pathways leading to frailty. The baseline biological changes associated with aging combined with subclinical disease from long-term “wear and tear” lead to the signs and symptoms of the frailty phenotype. Adapted with permission from Afilalo et al.7.
The overall prevalence of frailty in adults age 65 years and older has been estimated at approximately 10%. However, in patients with significant CVD, the prevalence may be as high as 60%.8,9 In 2009, Afilalo et al10 conducted a systematic review evaluating the risk of concomitant frailty and CVD. The study pooled 54 250 elderly patients from 9 studies and demonstrated an increased risk of death in those with concomitant frailty and CVD with an adjusted odds ratio (OR) ranging from 1.6 to 4.0 across the evaluated studies.
The frail phenotype is even more pervasive in valvular heart disease patients, particularly in calcific aortic stenosis (primarily seen in patients older than 70 years of age). In high risk transcatheter aortic valve implantation (TAVI) patients with calcific aortic stenosis, the prevalence of frailty was as high as 86%.11
FRAILTY PREDICTION SCORESA wide variety of frailty scores have been evaluated in the literature. These scores are usually based on variations of the 5 frailty markers originally described by Fried et al.1 The Fried frailty score encompasses a 5-point scale with a score of ≥ 3 being diagnostic of frailty. The components of the scale include slowness of gait (measured by a 5-meter speed test), physical strength (assessed by hand grip strength), physical activity levels, fatigue, and loss of body mass.1 An alternative test is the Short Physical Performance Battery (SPPB), which evaluates patients based on gait speed, chair rise strength, and balance. Each parameter of the SPPB is scored on a 0 to 4 scale, and a score of ≤ 5 of 12 is considered frail.12
FRAILTY IN CARDIAC SURGERY AND TRANSCATHETER VALVE PROCEDURESThe preponderance of literature evaluating frailty in CVD has focused on perioperative assessment prior to cardiac surgery and TAVI. Multiple studies have demonstrated that patients who are frail are at higher risk for morbidity and mortality after cardiac and noncardiac surgery.13 Makary et al14 showed a significant increased risk of postoperative complications (OR = 2.54; 95% confidence interval [95%CI], 1.12-5.77; P<.01), increased length of stay (incidence rate ratio 1.69; 95%CI, 1.28-2.23; P<.01), and need for postdischarge rehabilitation (OR = 20.48; 95%CI, 5.54-75.68; P<.01) in those patients who were deemed frail prior to general surgery. Lee et al15 evaluated frailty in cardiac surgery patients and found that frailty was associated with significantly increased odds of postoperative mortality (OR = 1.8; 95%CI, 1.1-3.0) and institutional discharge (OR = 6.3; 95%CI, 4.2-9.4).
In TAVI patients, frailty also carries an increased risk of mortality and postprocedural complications. Multiple small studies have evaluated frailty markers in patients undergoing TAVI (Table). In a study from our center evaluating 102 high-risk patients, approximately 80% were considered frail. A diagnosis of frailty was associated with increased mortality at 1 year (17% in the frail and 7% in the nonfrail: hazard ratio [HR] = 3.5; 95%CI, 1.4-8.5; P=.007) but was not associated with short-term events on multivariate analysis.21
Transcatheter Aortic Valve Implantation Trials Assessing Frailty
| Trial | No. | Design | Frailty model | Outcome |
|---|---|---|---|---|
| Osnabrugge et al16 | 471 | Multicenter prospective CoreValve® registry | Quality of life and functional scoresKCCQ, SF-12, EuroQol-5D | KCCQ increased at 12 months by 27.4 (95%CI, 24.1-30.8) |
| Yamamoto et al17 | 777 | Multicenter retrospective France-2 registry | BMI < 20 vs > 20 | No difference in 30-day or 1-year mortality or MACE |
| Puls et al18 | 300 | Single center prospective cohort study | Frailty assessment by Katz score | Frail patients had increased risk of mortalityImmediately post-TAVI: 5.5% vs 1.3%; P=.041-month post-TAVI: 17% vs 5.8%; P=.002 |
| Green et al4 | 484 | Multicenter prospective cohort(PARTNER trial) | 6-min walk speed | No difference in 30-day outcomesSlow walkers had improved walking distance after TAVI |
| Stortecky et al19 | 100 | Prospective cohort study | Frailty index score | Increased 1-year mortalityOR = 3.68; 95%CI, 1.21-11.19 |
| Schoenenberger et al20 | 119 | Prospective cohort study (same cohort as Stortecky et al19) | Frailty index score | Increased 1-year mortalityAdjusted OR = 1.56; 95%CI, 1.20-2.04; P=.001 |
| Green et al21 | 159 | Single center prospective cohort study | Gait speedGrip strengthAlbuminADLs | No difference at 1 month;Increased 1-year mortality, HR = 3.5; 95%CI, 1.4-9.5; P=.007 |
| Green et al22 | 102 | Single center prospective cohort study | Gait speed | No correlation between gait speed alone and adverse outcomes |
| Ewe et al23 | 147 | Multicenter prospective cohort study | Fried score > 3 | Increased 9-month mortality, HR = 4.2; 95%CI, 2.0-8.8 |
| Rodés-Cabau et al24 | 345 | Multicenter retrospective cohort study | Physician opinion | No difference in outcome at 30 days and 8 months |
95%CI, 95% confidence interval; ADLs, activities of daily living; BMI, body mass index; HR, hazard ratio; OR, odds ratio; KCCP, Kansas City Cardiomyopathy Questionnaire; MACE, major adverse cardiac events; SF-12, Short form 12; TAVI, transcatheter aortic valve implantation.
The University Medical Center of Göttingen in Germany recently published their experience with frail patients undergoing TAVI. Mortality was significantly increased in patients with frailty compared with the nonfrail (mortality immediately post-TAVI: 5.5% vs 1.3%; P=.04 and mortality 1 month post-TAVI: 17% vs 5.8%; P=.002). During long-term follow up (median 537 days), 56% of frail patients had died compared with 24% of nonfrail patients (P<.001).18
A third study evaluated risk factors for lack of improvement in “extreme risk” patients undergoing TAVI with the Medtronic CoreValve® system. Although the study showed an overall improvement in postprocedural symptoms and quality of life, a significant proportion of the patients (39%) did not experience improvement. Frailty markers associated with a lack improvement after TAVI were wheelchair bound status (OR = 2.6; 95%CI, 1.3-5.2; P=.006) and serum albumin < 3.3g/dL (OR = 1.8; 95%CI, 0.9-3.5; P=.073).16
These studies demonstrate the potential powerful impact of frailty on clinical event prediction in general surgery, cardiac surgery, and transcatheter procedures. However, the most commonly used risk scores for both cardiac and noncardiac surgery do not currently incorporate frailty in their metrics, ie, the Society for Thoracic Surgeons score and the Revised Cardiac Risk Index. The EuroSCORE II does have mobility as part of its risk calculation algorithm; however, it is arbitrarily defined as “severe impairment of mobility,” rather than being based on functional testing.25 The relative absence of frailty risk adjustment in these scores has led to questions regarding their predictive power elderly patients.13 The development of a TAVI specific risk score, which is likely to include frailty metrics, has been the subject of significant discussion but has not adopted to date.26
FRAILTY IN CORONARY ARTERY DISEASEStudies have demonstrated that frail, elderly individuals have a higher prevalence of coronary artery disease, a greater extent of angiographic disease (including left main disease), and carry a higher mortality than nonfrail patients.27,28 The EPESE (Established Populations for Epidemiologic Studies of the Elderly) study showed that impaired mobility was significantly associated with increased risk of death from coronary artery disease (adjusted relative risk: 1.9; 95%CI, 1.4-2.4).29
Frailty has also been associated with increased major adverse cardiac events after non—ST-segment elevation myocardial infarction. Ekerstad et al30 prospectively evaluated 307 patients with non—ST-segment elevation myocardial infarction. On multivariate analysis, frailty was strongly associated with major adverse cardiac events (adjusted OR = 2.2; 95%CI, 1.3-3.7) and adjusted mortality at 1 month (OR = 4.7; 95%CI, 1.7-13.0).
The “After Eighty Study” (NCT01255540)31 was recently presented at the 2015 American College of Cardiology conference. The study, which has not yet been published, randomized 457 patients over the age of 80 with non—ST-segment elevation myocardial infarction to invasive vs conservative strategy and found a reduction in major adverse cardiac events in the invasive arm (41% vs 61%; P<.001). This result indicates that, despite the higher mortality of elderly patients after non—ST-segment elevation myocardial infarction, older patients will still likely benefit from coronary revascularization in the correct clinical context. Subanalysis of this trial may give further guidance for the optimal treatment of non—ST-segment elevation myocardial infarction in frail patients over the age of 80 years.31
FRAILTY IN HEART FAILUREAs with coronary artery disease, frailty has been associated with higher rates of heart failure (HF) in the older population. Studies evaluating frailty and HF indicate that their coexistence results in greater overall morbidity, mortality, and hospitalization rates.7,32–34 The basic mechanisms underlying this risk may be, at least partly, due to a larger extent of myocardial injury occurring in response to stressors over time (ie, greater damage when demand ischemia occurs), which then leads to permanent myocardial injury, fibrosis, and more frequent clinical decompensation.
The Health ABC study evaluated the relationship between HF and frailty in a large cohort of adults aged 70 to 79 years over an 11-year period using the SPPB scale. The study found that, in comparison with nonfrail patients, frail individuals had a significant risk of developing HF (SPPB moderate frailty: HR = 1.36; 95%CI, 1.08-1.71; SPPB severe frailty: HR = 1.88; 95%CI, 1.02-3.47).35 Lupón et al32 evaluated patients with HF who were older than 70 years and found that 30% met frailty criteria at a younger than expected age, which was hypothesized to be due to overlap in the clinical syndromes of frailty and HF.7
A recent study by Sergi et al36 examined the concept of “pre-frailty” as a risk factor for the development of CVD. “Pre-frailty” was defined as the presence of 1 or 2 out of 5 positive factors on the Fried scale and was associated with a significantly higher risk of developing CVD. This result was driven primarily by new diagnoses of HF. Those patients meeting 2 criteria on the Fried frailty score were seen to have an 80% higher risk of developing CVD (HR = 1.79; 95%CI, 1.27-2.52; P=.001). Frailty parameters that were the best predictors of adverse events included low overall fitness level (HR = 1.70; 95%CI, 1.07-3.50; P=.03), fatigue (HR = 1.53; 95%CI, 1.09-2.14; P=.01), and slow gait speed (HR = 1.28; 95%CI, 1.03-1.71; P=.03).36 The FRAIL-HF trial37 is an ongoing prospective observational study that will further elucidate outcomes in risk factors in this population.
CONCLUSIONSThe impact of frailty on adverse clinical outcomes has been seen repeatedly across the spectrum of CVD. Frailty has been correlated with increased morbidity and mortality and decreased functional status in patients undergoing cardiac or noncardiac procedures. However, the quality of the available data on frailty is limited due to small sample sizes and a lack of randomized trials. Additionally, there is inadequate data comparing alternative frailty assessment tools, and significant debate remains as to which metrics are most clinically predictive.
Multiple studies are currently underway to address these issues. The FRAILTY-AVR study (NCT01845207) will evaluate the predictive ability of several frailty tools in aortic valve procedures. The impact of frailty on outcomes in older patients with acute coronary syndromes will be examined further in the SILVER-AMI trial (NCT01755052). Additionally, the Society for Thoracic Surgeons database is now collecting 5 meter gait speed information to allow for incorporation of this metric into their risk model.
Risk assessment and prognostication pose a significant challenge when facing complex decisions regarding cardiovascular management and potential use of invasive procedures in older patients. Frailty has become a valuable clinical tool correlated with a greater risk of myocardial infarction, HF, and death. Application of the current data and soon to be published clinical trial results will allow for further refinement of frailty assessment tools to facilitate enhanced shared decision-making between aging patients and their physicians.
CONFLICTS OF INTERESTP. Green receives grant support for frailty research from the National Institute of Health, Bethesda, Maryland, United States.