The prognostic value of chronic total occlusion in nonculprit coronary arteries in patients with myocardial infarction undergoing primary angioplasty remains controversial. Several publications have described different methodologies and conflicting findings. In addition, causes of death were not reported. Our aim is to analyze the prognostic impact of chronic total occlusion in nonculprit coronary arteries and the role of left ventricular ejection fraction in this analysis.
MethodsProspective inclusion of consecutive patients with ST-segment elevation myocardial infarction who underwent primary angioplasty. We recorded baseline characteristics, in-hospital clinical course, and mortality and its causes during follow-up. We assessed the impact of chronic total occlusion on mortality using Cox regression analysis.
ResultsChronic total occlusion in nonculprit arteries was present in 125 of 1176 patients (10.6%); in 79 of these 125 patients, chronic total occlusion was present in the proximal segments. The mean follow-up was 339 days; 64 (5.8%) patients died during the first 6 months. Patients with chronic total occlusions had more comorbidities, poorer ventricular function, and higher mortality (hazard ratio=2.79; 95% confidence interval, 1.71-4.56). Chronic total occlusion was also associated with noncardiac death (hazard ratio=3.83; 95% confidence interval, 2.10-7.01). Chronic total occlusion in proximal segments was associated with both cardiac (hazard ratio=3.22; 95% confidence interval, 1.42-7.30) and noncardiac deaths (hazard ratio=3.43; 95% confidence interval, 1.67-7.06). The multivariate analysis performed without including left ventricular ejection fraction showed a significant association between chronic total occlusion and mortality. However, when left ventricular ejection fraction was included in the analysis, this association was nonsignificant (hazard ratio=1.76; 95% confidence interval, 0.85-3.65; P=.166).
ConclusionsChronic total occlusion in this clinical setting identified patients at higher risk with more comorbidities and higher mortality, but did not behave as an independent predictor of mortality when left ventricular ejection fraction was included in the analysis.
Keywords
Primary angioplasty (PA) is currently the treatment of choice for acute myocardial infarction with persistent ST-segment elevation.1 In this clinical setting, the presence of multivessel disease is associated with poorer prognosis.2–4 However, the prognostic value of the presence of chronic total occlusion in nonculprit artery (CTOnr) in these patients is much more controversial.4–10 Although several previous articles have described a prognostic impact of CTOnr on mortality, more recent data have not confirmed this impact,4 the main methodological difference being the inclusion of variables such as left ventricular ejection fraction (LVEF) or creatinine clearance in the analyses. In addition, there is no information on the causes of death in this setting.
The purpose of our study was: a) to analyze the impact of CTOnr on patients’ total mortality 6 months post-PA in our setting and to elucidate the possible role of variables such as LVEF and Killip class in this association; b) to analyze the causes of mortality based on the presence of CTOnr, and c) to analyze the impact of the type of CTOnr vessel on mortality and its causes.
METHODSStudy Population and Health Care ProtocolAll patients with ST-segment elevation acute myocardial infarction referred to our hospital for PA in the first 12h postinfarction between October 2009 and June 2012 were prospectively included in the series. The reperfusion protocol is activated as part of the “Infarct Code” program implemented in the autonomous community of Catalonia in 2009. The criteria used to activate the protocol were the presence of chest pain with onset less than 12 h previously with ST-segment elevation of 1 mm or greater in 2 contiguous leads or newly acquired left bundle-branch block.
The therapeutic protocol included the administration at diagnosis of an oral dose of 250-300 mg of acetylsalicylic acid, an oral loading dose of 600 mg of clopidogrel, and parenteral anticoagulant, preferably with unfractionated heparin. Patients were immediately transferred to the referral interventional cardiology laboratory for emergent coronary angiography. Adjuvant antithrombotic therapy (bivalirudin, glycoprotein IIb/IIIa inhibitors) was administered at the operator's discretion. The percutaneous intervention techniques, choice of stent type, and drug therapy during and after catheterization were at the operator's discretion and in accordance with current recommendations.11,12
Nonculprit coronary lesions were revascularized during the initial angiography only in patients with frank hemodynamic or electric instability. Nonculprit coronary lesions were revascularized during hospitalization or follow-up at the discretion of the medical team in charge, based on the patient's clinical progress, ventricular function, degree of inducible ischemia, and angiographic features of documented lesions.
Definitions and Data CollectionTrained cardiologists collected the data prospectively, using a standard form. The data recorded consisted of baseline characteristics, medical history, biochemical and electrocardiographic findings, echocardiographic and angiographic parameters, procedures performed, in-hospital treatments, complications, and in-hospital mortality.
The hemodynamic parameters (heart rate, systolic blood pressure) and Killip class were recorded when the patient was admitted to the coronary unit. Creatinine clearance was calculated by the Cockcroft-Gault formula.13
LVEF was analyzed as of day 3 postinfarction by transthoracic echocardiogram using the Simpson method.
Coronary disease was quantitated by taking into account the number of epicardial arterial territories (anterior interventricular, circumflex, right coronary) with artery lumen stenosis of 70% or greater, or 50% in the case of the left common trunk. The degree of stenosis was quantitated by visual analysis. Chronic total occlusion was considered to be total (100%) occlusion of the artery lumen without anterograde flow or with flow (anterograde or retrograde) through collateral vessels in an artery other than the culprit artery.5 The differentiation between CTOnr and acute occlusion was likewise based on the morphologic analysis (absence of fresh thrombus, presence of well-developed collateral circulation or microchannels) by the interventional cardiologist who performed the procedure.14 CTOnr in a proximal artery was considered to be present in the case of segments 1-3 (right coronary), 6-7 (anterior interventricular), and 11-12 (circumflex) according to the CASS classification.15
Primary and Secondary EndpointsThe primary endpoint of the study was total 6-month mortality (including in-hospital mortality). As secondary endpoints, we recorded 6-month cardiac and noncardiac mortality.
Vital status information was collected by an analysis of hospital records and by telephone contact with the patients’ relatives or treating physician. The cause of death was determined by the medical practitioner who attended the patient at the time of death. If death occurred outside the hospital setting, the cause was determined by an interview with relatives. If there was more than 1 cause of death, the main cause was established by considering the clinical relevance of each one. Cardiac death was considered to be any death due to myocardial infarction or heart failure and sudden death.
Statistical AnalysisPASW Statistics 18 (Chicago, Illinois, United States) was used for the data analysis. The categorical variables are expressed as number and percentage, and the quantitative variables are expressed as mean (standard deviation). Variables with a nonnormal distribution are expressed as median [interquartile range]. The normality of distributions was analyzed by the Kolmogorov-Smirnoff test.
The categorical variables were compared by the χ2 test or Fisher's exact test when applicable. The quantitative variables were compared by the Student t test. Survival curves were obtained by the Kaplan-Meier method.
Analysis of the Relationship Between Chronic Total Occlusion in Nonculprit Artery and MortalityMortality related to CTOnr was analyzed by the Cox regression method, and the proportional hazard model assumption was confirmed by the Kalbfleisch and Prentice method.16 Potential confounders included in the multivariate analysis had to meet the following requirements17: statistically significant association (P≤.2) with both exposure (CTOnr) and effect (mortality); clinically reasonable potential confounding effect between CTOnr and mortality, and not an intermediate variable in the relationship between CTOnr and mortality.
The variables of LVEF and Killip class on admission were handled in a specific manner, as they could be considered, at least partly, as intermediate variables between CTOnr and mortality. However, because they are clinically relevant, multivariate analyses were performed with and without these 2 variables to study the association between CTOnr and mortality.
The association between CTOnr and mortality in the final model was considered statistically significant if the hazard ratio (HR) resulted in P≤.05 and its 95% confidence interval (95%CI) did not include the value of 1.
Analysis of the Relationship Between Chronic Total Occlusion in Nonculprit Artery in Proximal Arteries and MortalityTo analyze the relationship between CTOnr in proximal arteries and mortality, the same statistical procedure was followed as with all CTOnr.
RESULTSDuring the period analyzed, 1176 patients were referred to our hospital for PA; of these, 125 (10.6%) had CTOnr in at least 1 coronary artery, 79 of whom had a CTOnr in at least 1 proximal artery.
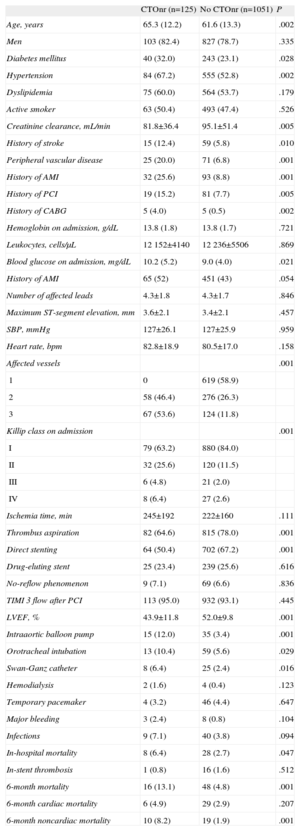
The patients’ baseline characteristics, procedures, and clinical course according to the presence of CTOnr are listed in Table 1. Patients with CTOnr were older and had a higher burden in terms of cardiovascular risk factors and other comorbidities. They were also more likely to show signs of heart failure, more massive coronary disease, and worse ventricular function at discharge. Their requirements for invasive procedures during hospitalization (intraaortic balloon pump, invasive mechanical ventilation, Swan-Ganz catheter) were significantly higher than those of patients without CTOnr.
Baseline Clinical Characteristics, Procedures, and Complications According to the Presence of Chronic Total Occlusion in Nonculprit Artery
| CTOnr (n=125) | No CTOnr (n=1051) | P | |
| Age, years | 65.3 (12.2) | 61.6 (13.3) | .002 |
| Men | 103 (82.4) | 827 (78.7) | .335 |
| Diabetes mellitus | 40 (32.0) | 243 (23.1) | .028 |
| Hypertension | 84 (67.2) | 555 (52.8) | .002 |
| Dyslipidemia | 75 (60.0) | 564 (53.7) | .179 |
| Active smoker | 63 (50.4) | 493 (47.4) | .526 |
| Creatinine clearance, mL/min | 81.8±36.4 | 95.1±51.4 | .005 |
| History of stroke | 15 (12.4) | 59 (5.8) | .010 |
| Peripheral vascular disease | 25 (20.0) | 71 (6.8) | .001 |
| History of AMI | 32 (25.6) | 93 (8.8) | .001 |
| History of PCI | 19 (15.2) | 81 (7.7) | .005 |
| History of CABG | 5 (4.0) | 5 (0.5) | .002 |
| Hemoglobin on admission, g/dL | 13.8 (1.8) | 13.8 (1.7) | .721 |
| Leukocytes, cells/μL | 12152±4140 | 12236±5506 | .869 |
| Blood glucose on admission, mg/dL | 10.2 (5.2) | 9.0 (4.0) | .021 |
| History of AMI | 65 (52) | 451 (43) | .054 |
| Number of affected leads | 4.3±1.8 | 4.3±1.7 | .846 |
| Maximum ST-segment elevation, mm | 3.6±2.1 | 3.4±2.1 | .457 |
| SBP, mmHg | 127±26.1 | 127±25.9 | .959 |
| Heart rate, bpm | 82.8±18.9 | 80.5±17.0 | .158 |
| Affected vessels | .001 | ||
| 1 | 0 | 619 (58.9) | |
| 2 | 58 (46.4) | 276 (26.3) | |
| 3 | 67 (53.6) | 124 (11.8) | |
| Killip class on admission | .001 | ||
| I | 79 (63.2) | 880 (84.0) | |
| II | 32 (25.6) | 120 (11.5) | |
| III | 6 (4.8) | 21 (2.0) | |
| IV | 8 (6.4) | 27 (2.6) | |
| Ischemia time, min | 245±192 | 222±160 | .111 |
| Thrombus aspiration | 82 (64.6) | 815 (78.0) | .001 |
| Direct stenting | 64 (50.4) | 702 (67.2) | .001 |
| Drug-eluting stent | 25 (23.4) | 239 (25.6) | .616 |
| No-reflow phenomenon | 9 (7.1) | 69 (6.6) | .836 |
| TIMI 3 flow after PCI | 113 (95.0) | 932 (93.1) | .445 |
| LVEF, % | 43.9±11.8 | 52.0±9.8 | .001 |
| Intraaortic balloon pump | 15 (12.0) | 35 (3.4) | .001 |
| Orotracheal intubation | 13 (10.4) | 59 (5.6) | .029 |
| Swan-Ganz catheter | 8 (6.4) | 25 (2.4) | .016 |
| Hemodialysis | 2 (1.6) | 4 (0.4) | .123 |
| Temporary pacemaker | 4 (3.2) | 46 (4.4) | .647 |
| Major bleeding | 3 (2.4) | 8 (0.8) | .104 |
| Infections | 9 (7.1) | 40 (3.8) | .094 |
| In-hospital mortality | 8 (6.4) | 28 (2.7) | .047 |
| In-stent thrombosis | 1 (0.8) | 16 (1.6) | .512 |
| 6-month mortality | 16 (13.1) | 48 (4.8) | .001 |
| 6-month cardiac mortality | 6 (4.9) | 29 (2.9) | .207 |
| 6-month noncardiac mortality | 10 (8.2) | 19 (1.9) | .001 |
AMI, acute myocardial infarction; CABG, coronary aortic bypass graft; CTOnr, chronic total occlusion in nonculprit artery; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention; SBP, systolic blood pressure; TIMI, Thrombolysis In Myocardial Infarction.
The data are presented as no. (%) or mean± standard deviation.
There were no significant differences in ischemia time or TIMI (Thrombolysis In Myocardial Infarction) flow in the culprit artery at the end of the interventional procedure. In-hospital mortality was significantly greater in the patients with CTOnr, but no significant differences were observed in the in-hospital incidence of infectious and hemorrhagic complications. No differences were observed in the incidence of reinfarction (3.5% vs 3.4%; P=.977) or the need for new revascularization (7.8% vs 8.9%; P=.693) during follow-up. In 4 (3.2%) patients, the CTOnr was revascularized during follow-up.
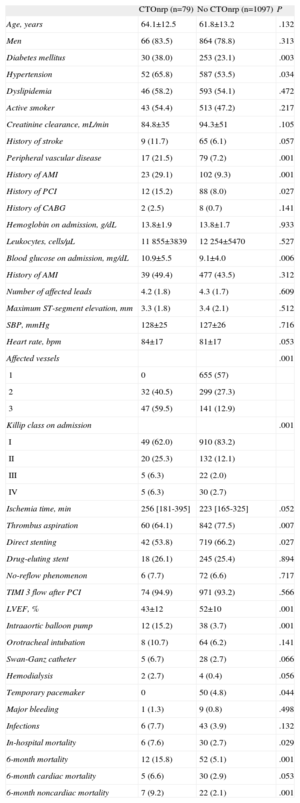
The comparative analysis of patients with CTOnr in proximal arteries compared with all others showed similar findings to those observed in all patients with CTOnr (Table 2); although these patients had higher comorbidity burdens, more severe coronary disease, worse ventricular function at discharge, and higher in-hospital mortality, no significant differences were found in the incidence of infectious and hemorrhagic complications. The ischemia time was slightly higher in patients with CTOnr in proximal segments, although this different was not statistically significant.
Baseline Clinical Characteristics, Procedures, and Complications According to the Presence of Chronic Total Occlusion in Nonculprit Artery in Proximal Segments
| CTOnrp (n=79) | No CTOnrp (n=1097) | P | |
| Age, years | 64.1±12.5 | 61.8±13.2 | .132 |
| Men | 66 (83.5) | 864 (78.8) | .313 |
| Diabetes mellitus | 30 (38.0) | 253 (23.1) | .003 |
| Hypertension | 52 (65.8) | 587 (53.5) | .034 |
| Dyslipidemia | 46 (58.2) | 593 (54.1) | .472 |
| Active smoker | 43 (54.4) | 513 (47.2) | .217 |
| Creatinine clearance, mL/min | 84.8±35 | 94.3±51 | .105 |
| History of stroke | 9 (11.7) | 65 (6.1) | .057 |
| Peripheral vascular disease | 17 (21.5) | 79 (7.2) | .001 |
| History of AMI | 23 (29.1) | 102 (9.3) | .001 |
| History of PCI | 12 (15.2) | 88 (8.0) | .027 |
| History of CABG | 2 (2.5) | 8 (0.7) | .141 |
| Hemoglobin on admission, g/dL | 13.8±1.9 | 13.8±1.7 | .933 |
| Leukocytes, cells/μL | 11 855±3839 | 12 254±5470 | .527 |
| Blood glucose on admission, mg/dL | 10.9±5.5 | 9.1±4.0 | .006 |
| History of AMI | 39 (49.4) | 477 (43.5) | .312 |
| Number of affected leads | 4.2 (1.8) | 4.3 (1.7) | .609 |
| Maximum ST-segment elevation, mm | 3.3 (1.8) | 3.4 (2.1) | .512 |
| SBP, mmHg | 128±25 | 127±26 | .716 |
| Heart rate, bpm | 84±17 | 81±17 | .053 |
| Affected vessels | .001 | ||
| 1 | 0 | 655 (57) | |
| 2 | 32 (40.5) | 299 (27.3) | |
| 3 | 47 (59.5) | 141 (12.9) | |
| Killip class on admission | .001 | ||
| I | 49 (62.0) | 910 (83.2) | |
| II | 20 (25.3) | 132 (12.1) | |
| III | 5 (6.3) | 22 (2.0) | |
| IV | 5 (6.3) | 30 (2.7) | |
| Ischemia time, min | 256 [181-395] | 223 [165-325] | .052 |
| Thrombus aspiration | 60 (64.1) | 842 (77.5) | .007 |
| Direct stenting | 42 (53.8) | 719 (66.2) | .027 |
| Drug-eluting stent | 18 (26.1) | 245 (25.4) | .894 |
| No-reflow phenomenon | 6 (7.7) | 72 (6.6) | .717 |
| TIMI 3 flow after PCI | 74 (94.9) | 971 (93.2) | .566 |
| LVEF, % | 43±12 | 52±10 | .001 |
| Intraaortic balloon pump | 12 (15.2) | 38 (3.7) | .001 |
| Orotracheal intubation | 8 (10.7) | 64 (6.2) | .141 |
| Swan-Ganz catheter | 5 (6.7) | 28 (2.7) | .066 |
| Hemodialysis | 2 (2.7) | 4 (0.4) | .056 |
| Temporary pacemaker | 0 | 50 (4.8) | .044 |
| Major bleeding | 1 (1.3) | 9 (0.8) | .498 |
| Infections | 6 (7.7) | 43 (3.9) | .132 |
| In-hospital mortality | 6 (7.6) | 30 (2.7) | .029 |
| 6-month mortality | 12 (15.8) | 52 (5.1) | .001 |
| 6-month cardiac mortality | 5 (6.6) | 30 (2.9) | .053 |
| 6-month noncardiac mortality | 7 (9.2) | 22 (2.1) | .001 |
AMI, acute myocardial infarction; CABG, coronary aortic bypass graft; CTOnr, chronic total occlusion in nonculprit artery in proximal segments; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention; SBP, systolic blood pressure; TIMI, Thrombolysis In Myocardial Infarction.
The data are presented as no. (%), mean± standard deviation or median [interquartile range].
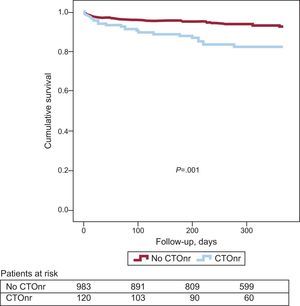
Follow-up data were collected from 1112 (94.6%) patients, 122 (97.6%) in the group with CTOnr and 990 (94.1%) in the group without CTOnr. The mean follow-up was 339 days. Total mortality during follow-up was significantly higher among patients with CTOnr (HR=2.79; 95%CI, 1.71-4.56; P<.001; 6-month mortality among patients with CTOnr, 16 of 122 [13.1%]; 6-month mortality among patients without CTOnr, 48 of 990 [4.8%]). When the causes of death were analyzed separately, a statistically significant association was observed between CTOnr and noncardiac mortality (HR=3.83; 95%CI, 2.10-7.01; P<.001). The association between CTOnr and cardiac mortality was not statistically significant (HR=1.86; 95%CI, 0.82-4.21; P=.138; 6-month cardiac mortality among patients with nonculprit, 6 of 122 [4.9%]; 6-month cardiac mortality among patients without CTOnr, 29 of 990 [2.9%]). Figure 1 shows the trend for total cumulative mortality according to the presence of CTOnr.
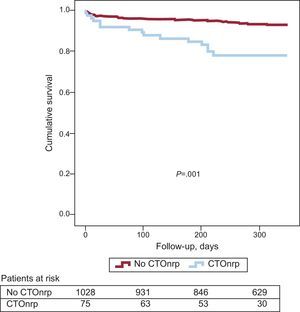
Association Between Chronic Total Occlusion in Nonculprit Artery in Proximal Segments and MortalityThe results of this analysis were very similar to the previous results. Total mortality during follow-up was significantly higher among patients with CTOnr in proximal segments (HR=3.18; 95%CI, 1.82-5.55; P<.001). However, unlike the previous analysis, the association between CTOnr and mortality was statistically significant for both cardiac (HR=3.22; 95%CI, 1.42-7.30; P=.005) and noncardiac causes (HR=3.43; 95%CI, 1.67-7.06; P<.001). Figure 2 shows the trend for total cumulative mortality according to the presence of CTOnr in proximal segments.
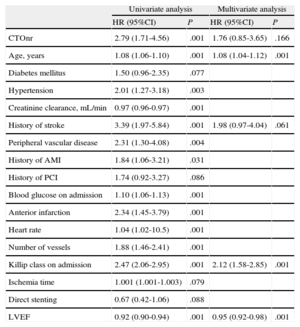
Multivariate AnalysisRelationship Between Chronic Total Occlusion in Nonculprit Artery and MortalityThe multivariate analysis performed without including LVEF or Killip class on admission showed a significant association between CTOnr and mortality (HR=2.09; 95%CI, 1.16-3.77; P=.014). When both variables were included in the analysis, the statistical model showed a nonsignificant association (HR=1.76; 95%CI, 0.85-3.65; P=.166). Table 3 lists the results of the univariate and multivariate analyses, including all potential confounders.
Analysis of the Relationship Between Chronic Total Occlusion in Nonculprit Artery and Mortality
| Univariate analysis | Multivariate analysis | |||
| HR (95%CI) | P | HR (95%CI) | P | |
| CTOnr | 2.79 (1.71-4.56) | .001 | 1.76 (0.85-3.65) | .166 |
| Age, years | 1.08 (1.06-1.10) | .001 | 1.08 (1.04-1.12) | .001 |
| Diabetes mellitus | 1.50 (0.96-2.35) | .077 | ||
| Hypertension | 2.01 (1.27-3.18) | .003 | ||
| Creatinine clearance, mL/min | 0.97 (0.96-0.97) | .001 | ||
| History of stroke | 3.39 (1.97-5.84) | .001 | 1.98 (0.97-4.04) | .061 |
| Peripheral vascular disease | 2.31 (1.30-4.08) | .004 | ||
| History of AMI | 1.84 (1.06-3.21) | .031 | ||
| History of PCI | 1.74 (0.92-3.27) | .086 | ||
| Blood glucose on admission | 1.10 (1.06-1.13) | .001 | ||
| Anterior infarction | 2.34 (1.45-3.79) | .001 | ||
| Heart rate | 1.04 (1.02-10.5) | .001 | ||
| Number of vessels | 1.88 (1.46-2.41) | .001 | ||
| Killip class on admission | 2.47 (2.06-2.95) | .001 | 2.12 (1.58-2.85) | .001 |
| Ischemia time | 1.001 (1.001-1.003) | .079 | ||
| Direct stenting | 0.67 (0.42-1.06) | .088 | ||
| LVEF | 0.92 (0.90-0.94) | .001 | 0.95 (0.92-0.98) | .001 |
95%CI, 95% confidence interval; AMI, acute myocardial infarction; CTOnr, chronic total occlusion in nonculprit artery; HR, hazard ratio; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention.
Univariate analysis of the association of each potential confounder with mortality. The multivariate analysis includes all potential confounders (variables with statistical association P<.2 with exposure [chronic total occlusion in nonculprit artery; Table 1] and effect [mortality; univariate analysis in this same table], including left ventricular ejection fraction and Killip class on admission).
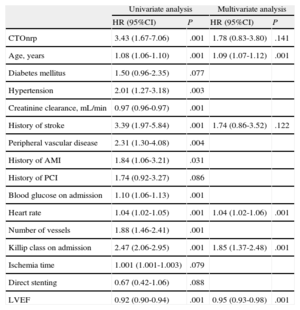
The results of this analysis were very similar to those of the previous analysis. The multivariate analysis performed without including LVEF or Killip class on admission showed a significant association between CTOnr in proximal segments and mortality (HR=2.53; 95%CI, 1.29-4.96; P=.007). Again, when both variables were included, the association observed was no longer statistically significant (HR=1.78; 95%CI, 0.83-3.80; P=.141). Table 4 lists the results of the univariate and multivariate analyses, including all potential confounders.
Analysis of the Relationship Between Chronic Total Occlusion in Nonculprit Artery in Proximal Segments and Mortality
| Univariate analysis | Multivariate analysis | |||
| HR (95%CI) | P | HR (95%CI) | P | |
| CTOnrp | 3.43 (1.67-7.06) | .001 | 1.78 (0.83-3.80) | .141 |
| Age, years | 1.08 (1.06-1.10) | .001 | 1.09 (1.07-1.12) | .001 |
| Diabetes mellitus | 1.50 (0.96-2.35) | .077 | ||
| Hypertension | 2.01 (1.27-3.18) | .003 | ||
| Creatinine clearance, mL/min | 0.97 (0.96-0.97) | .001 | ||
| History of stroke | 3.39 (1.97-5.84) | .001 | 1.74 (0.86-3.52) | .122 |
| Peripheral vascular disease | 2.31 (1.30-4.08) | .004 | ||
| History of AMI | 1.84 (1.06-3.21) | .031 | ||
| History of PCI | 1.74 (0.92-3.27) | .086 | ||
| Blood glucose on admission | 1.10 (1.06-1.13) | .001 | ||
| Heart rate | 1.04 (1.02-1.05) | .001 | 1.04 (1.02-1.06) | .001 |
| Number of vessels | 1.88 (1.46-2.41) | .001 | ||
| Killip class on admission | 2.47 (2.06-2.95) | .001 | 1.85 (1.37-2.48) | .001 |
| Ischemia time | 1.001 (1.001-1.003) | .079 | ||
| Direct stenting | 0.67 (0.42-1.06) | .088 | ||
| LVEF | 0.92 (0.90-0.94) | .001 | 0.95 (0.93-0.98) | .001 |
95%CI, 95% confidence interval; AMI, acute myocardial infarction; CTOnrp, chronic total occlusion in nonculprit artery in proximal segments; HR, hazard ratio; LVEF, left ventricular ejection fraction; PCI, percutaneous coronary intervention.
Univariate analysis of association of each potential confounder with P<.2 with mortality. The multivariate analysis includes all potential confounders (variables with statistical association [P<.2] with exposure [chronic total occlusion in nonculprit artery in proximal segments; Table 2] and effect [mortality; univariate analysis in this same table], including left ventricular ejection fraction and Killip class on admission).
The main findings of our study were: a) CTOnr represented a special-risk subgroup, with more comorbidities and poorer clinical course; b) this worse prognosis was mainly due to noncardiac mortality in all patients with CTOnr, and c) the presence of CTOnr did not behave in our series as an independent predictor of mortality when LVEF was included in the analyses.
The prevalence of CTOnr in our series was similar to that of previously published series, which reported levels of 8% to 13%.4,7–10 The characteristics of patients with CTOnr were also similar to those of earlier series,4,7,8 with older ages and greater comorbidity burdens, severity of coronary disease, ventricular dysfunction, and medium-term mortality.
The prognostic implication of CTOnr in patients who undergo PA is controversial. The observation of a worse prognosis attributable to the CTOnr in this setting could further justify early and careful reperfusion of the culprit artery as well as more intensive therapeutic management in the acute phase, in order to manage potential complications earlier and while the patient is more clinically stable.
Although many articles that have addressed this question describe an independent prognostic value of the presence of CTOnr,5–8,10 recent data do not support this hypothesis.4 The populations analyzed are different (in some cases, from clinical trials7,8) and there may be major methodological differences, as most authors do not include LVEF in the analyses. In addition, the severity of the coronary disease was coded differently.
Recently, Bataille et al.4 analyzed a population of 2020 consecutive patients who underwent PA, concluding that the worse prognosis of patients with CTOnr was mainly caused by the higher comorbidity burden and severity of coronary disease, rather than by the actual presence of CTOnr. The main difference between this study and earlier studies is the inclusion of LVEF and baseline glomerular filtration rate in the analysis of the association between CTOnr and mortality.
The inclusion of LVEF in this analysis is controversial. The presence of CTOnr in an artery contralateral to the culprit artery in patients with ST-segment elevation acute myocardial infarction could lead to worse collaterality in the acute phase and, consequently, larger infarction and worse residual LVEF. In fact, the presence of CTOnr has been correlated with worse ventricular function after infarction.6 Likewise, the presence of CTOnr could explain a worse Killip class on admission in this clinical setting. Consequently, it could be considered that both variables are, at least partly, intermediate in the relationship between CTOnr and mortality, and their inclusion in the analyses could contribute to an underestimation of the association between exposure and effect (type II statistical error).
On the other hand, noninclusion of LVEF because it is part of the CTOnr-mortality causal chain could result in an opposite bias. Patients with CTOnr clearly had more adverse characteristics (older age, more severe coronary disease, more frequent history of infarction) that could explain some degree of preexisting ventricular dysfunction. Therefore, excluding LVEF could theoretically magnify the association between chronic total occlusion and mortality. This reasoning, along with the fact that LVEF inclusion in the analyses is the main difference between the negative result obtained by Bataille et al.4 and the positive results obtained in previous studies, was what led the authors to perform analyses with and without these 2 variables, in an attempt to further analyze the relationship between CTOnr and mortality.
The results do not clearly indicate a prognostic implication of the CTOnr in this setting. The statistical significance of the association between CTOnr and mortality was observed only after exclusion of powerful mortality predictors such as LVEF or Killip class. Although both factors may be part of the causal chain between exposure and effect, excluding variables of such clinical relevance and such clear prognostic implications (largely unrelated to the presence of CTOnrs) could be hard to justify.
An analysis of the causes of mortality (not available in previous series) also provides relevant information. Higher noncardiac mortality in patients with CTOnr as a whole would support the hypothesis of CTOnr as a marker of total and comorbidity risk, rather than as a predictor of cardiac mortality per se. A CTOnr in a proximal artery could have a somewhat different prognostic implication. The larger myocardial area at risk could explain the higher cardiac mortality in these patients. Earlier series are also inconsistent in these aspects, as some restrict their analysis to CTOnr in arteries of a certain size,5 with technically feasible revascularization, or affecting a significant myocardial area,4 while other series include all patients with CTOnr.
The possible benefit of CTOnr revascularization in this clinical setting is controversial. Some data might support this strategy. A recent nonrandomized study pointed out the benefits of successful revascularization of the CTOnr in 136 patients 7 to 10 days after PA for ST-segment elevation acute myocardial infarction.18 Nonetheless, the paucity of information means that randomized trial data19 are needed to answer this question adequately.
LimitationsOur study has several limitations. Firstly, the study consisted of a single-center observational registry with a relatively low number of patients with CTOnr. The low rate of events limits the power of the predictive model. In patients who died before day 3, the last LVEF before death was recorded, but the authors felt that any resulting bias would be irrelevant. Because of the low rate of revascularization of CTOnrs in our patients (findings also described in the literature20), it was impossible to analyze the possible contribution of this variable to the clinical course. Nevertheless, earlier series also did not include this variable in their analyses. Lastly, because this is a relatively recent series, some patients had a somewhat shorter follow-up time than that reported in some of the published series.
CONCLUSIONSIn unselected patients with ST-segment elevation acute myocardial infarction who underwent PA, a CTOnr identified a group with a higher risk profile and more comorbidities. These patients had higher total medium-term mortality, particularly due to noncardiac causes. When LVEF was included in the analyses, CTOnr did not behave as an independent predictor of mortality in this setting.
CONFLICTS OF INTERESTNone declared.