Keywords
INTRODUCTION
Since its introduction in clinical practice 40 years ago, coronary artery bypass grafting (CABG) is a well-recognized treatment for stable coronary artery disease, with a major impact on survival.1
Long-term follow-up studies report 67% survival free of myocardial infarction (MI) 20 years after CABG.2 More recent data indicated an incidence of MI of 2% to 3% per year over the first 5 years, with a 5-year cumulative incidence of non-fatal MI of 15%,3 and a recurrent infarction in as many as 36% of patients at 10 years.4 Furthermore, CABG has a positive influence on long-term quality of life and functional capacity.5
On the other hand, observational studies on acute coronary syndromes (ACS) noted an overall prevalence of 20% for patients with a history of CABG.6,7
In these patients, there are 2 potential explanations, not mutually exclusive, for a new major coronary event, namely, progression of atherosclerotic disease in native arteries or the development of vascular disease in the grafts. Previous authors of an angiography study reported 8% venous graft failure at 1 year, 38% at 5 years, and 75% at 10 years after surgery,8 and progression of the atherosclerotic process in up to 51% 15 years after CABG.9
The prognosis of this subset of patients, nevertheless, remains doubtful. The Thrombolysis in Myocardial Infarction (TIMI) III ACS subset registry found a similar result during the hospital stay and at 6 months; however, by 1 year the subgroup of patients with a history of CABG had a poorer prognosis (incidence of combined events, 39.3% vs 30.2%; P=.002).10 Despite the poorer outcome, previous CABG was not an independent predictor of death in TIMI,11 Global Registry of Acute Coronary Events (GRACE),12 or PURSUIT13 scores. In contrast, several small series suggest a more favorable outcome for this group of patients, for both MI14 and unstable angina (UA).15
Although no randomized studies have been designed to evaluate the benefits of an invasive versus conservative strategy in patients who have already undergone CABG, recent guidelines recommend a low threshold for angiography with a preferable intervention in native circulation, and medical treatment similar to that used in the overall population (class I recommendation, level of evidence C).16
Due to the lack of data for this important subgroup of ACS patients when compared to others, we decided to perform to an analysis of the in-hospital and medium-term prognosis of an unselected ACS population with previous CABG from a single center.
METHODS
A continuous, observational, prospective study of 1495 consecutive admissions for ACS between May 2004 and December 2006 was conducted in a single coronary care unit. We excluded 130 patients because they were readmissions, and an additional 33 due to incomplete data. The final population consisted of 1296 patients.
ST-elevation AMI (STEMI) was defined as ischemic chest pain associated with new onset ST elevation greater than 1 mm in the ECG in at least 2 contiguous leads.
Non-ST AMI (NSTEMI) was synonymous with ischemic chest pain lasting more than 5 minutes, and positive cardiac biomarkers (troponin I) with or without ECG ischemic changes (eg, ST depression or T-wave inversion).
UA was defined as new onset angina (at least class III Canadian Cardiovascular Society [CCS]), progressive angina, or angina at rest, with or without ECG ischemic changes.
All patients gave written informed consent for participation, and the local ethics committee approved the study.
We analyzed standard records during admission, including the following data: demographic, clinical, electrical, echocardiography, laboratory, medical therapy, catheterization, in-hospital course, and discharge medication.
Median clinical follow-up after hospital discharge was 19 months. The information was collected by phone, at the outpatient clinic, or from hospital records in the event of a local repeated hospital admission. The primary endpoint was all-cause mortality and the combined result of cardiovascular death, nonfatal infarction, readmission for UA, and percutaneous coronary intervention as major adverse cardiac events (MACE). We also recorded MI, readmission for heart failure, and stroke.
Statistical Analysis
Continuous data are presented as the median [interquartile range] and compared using the Mann-Whitney test, or as the mean [SD], and compared using the Student t test. Categorical variables are reported as frequencies and percentages, and the c2 test or the exact Fisher test was used when appropriate.
The cumulative survival curves were constructed using the Kaplan-Meier methods, and the groups were compared by the log-rank test. The observational period started at hospital discharge and ended 1 year after the ACS.
A Cox regression analysis was performed for the clinical endpoint of UA 1 year after the ACS. The variables included in the model were significant at P less than .05 in the univariate analysis and included a history of CABG, diabetes, age over 75 years, STEMI, left ventricular ejection fraction (LVEF) lower than 40%, and invasive strategy.
All statistical tests were 2-tailed, and a P less than .05 was considered significant. All analyses were performed using SPSS 15 (Statistical Package for Social Sciences, SPSS Inc, Chicago, Illinois, United States).
RESULTS
The population was divided into 2 groups based on a history of CABG. Group A included 73 patients with previous CABG and group B, 1223 with no previous history of myocardial revascularization surgery.
Baseline Characteristics
The study cohort included 908 men and 388 women with a mean age of 67.0 (12.6) years; 5.6% had a history of CABG.
The baseline demographic and clinical characteristics of both groups are shown in Tables 1 and 2.
Patients with a history of CABG were more likely to be male, more often admitted for NSTEMI and UA, and more likely to have a history of diabetes, previous infarction, and PCI.
Left bundle-branch block and a higher TIMI risk score were more frequently associated with a history of CABG, as was a lower LVEF (50.0% [42.0%-55.0%] vs 53.0% [46.75%-60%], P<.001). In contrast, a higher peak troponin I was present in group B patients.
Previous History
The present ACS that prompted inclusion in the study occurred a median of 9 (4-12.5) years after CABG. Multivessel coronary artery disease was the most frequent coronary anatomic abnormality before surgery, and 13% and 37% of patients had lesions in the left main artery and proximal left anterior descending artery, respectively.
Arterial bypasses (only mammary internal bypass were performed) were more common than venous bypasses, and 83.3% of patients were treated by both types. Triple (38.9%) and double (37.0%) bypasses were the most common option (Table 3). All patients were operated with classic on-pump surgery.
In-Hospital Management
An invasive strategy was used in 38 (52.1%) CABG patients, compared to 732 (59.9%) non-CABG patients. Despite these absolute differences, both groups had a similar probability of receiving invasive treatment. There were no significant differences regarding medical treatment during the first 24 hours (Table 4).
Regarding catheterization laboratory data after admission for ACS, patients with a history of CABG had, as expected, more extensive coronary involvement (Table 5).
In 50% of patients with previous CABG, we concluded that graft lesions were contributing to the ACS. PCI was performed in 73.2% (460/628) of patients with no history of CABG, compared to 47.1% (16/34) of those who did (P=.001). Group B patients were also more likely to be fully revascularized and treated with a stent. Nevertheless, the odds for receiving a drug-eluting stent (DES) were similar in both groups. No patient in group A underwent a second surgical revascularization procedure.
We analyzed 55 venous grafts in the 38 patients admitted for an invasive procedure. There were 54.2% total occlusions, and most lesions were in the ostial and proximal segments of the grafts. In the case of arterial bypass grafts, a total of 30 grafts were analyzed. Significant atherosclerotic lesions were identified in only 6; half of these also had total occlusions.
PCI was performed in 7 patients in the venous grafts (Table 6).
Despite the lower rate of dual antiplatelet therapy, at discharge there were no other differences between groups with regard to aspirin, statin, ACE inhibitor, or beta blocker therapy.
Follow-up
In-hospital mortality was similar in both groups (9.6% vs 5.9% P=.20). Loss to follow-up was 6.3%; hence, data on the primary endpoint (all-cause mortality at 1 year) were available for 1140 patients.
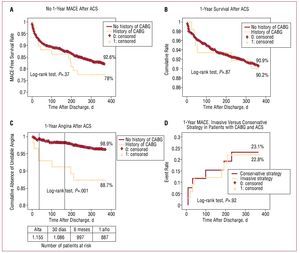
No differences in 30-day, 6-month, or 1-year all-cause mortality were detected (Figure 1B). The incidence of 1-year MACE was also similar (22.0% vs 17.4%, P=.37) (Figure 1A); the result was also similar for readmissions for heart failure or MI (fatal or nonfatal) (Table 7).
Figure. A: Kaplan-Meyer curves for cumulative MACE-free survival for ACS patients (history of CABG vs no history of CABG). B: Kaplan-Meyer curves showing overall survival for ACS patients (history of CABG vs no history of CABG). C: Kaplan-Meyer curves showing cumulative UA-free admission for ACS patients (history of CABG vs no history of CABG). D: Kaplan-Meyer curves showing 1-year MACE for patients with a history of CABG, admitted for an invasive or conservative strategy. ACS indicates acute coronary syndrome, CABG, coronary artery bypass graft; MACE, major adverse cardiac events.
The 1-year readmission rate for UA was significantly higher for patients with a history of CABG (11.3% vs 3.1%, P=.001) (Figure 1C), and the results remained significant in the multivariate Cox regression analysis (hazard ratio [HR] =4.49; 95% confidence interval [CI], 1.7-12.1), in a model that included the following variables: previous CABG, diabetes (HR=1.76; 95% CI, 0.80-3.87), age over 75 years (HR=0.98; 95% CI, 0.41-2.34), left ventricular ejection fraction lower than 40% (HR=0.80; 95% CI, 0.27-2.36), and invasive strategy (HR=0.84; 95% CI, 0.37-1.91) (overall χ2, 14.9).
In the previous CABG group, an invasive strategy during the acute phase did not influence 1-year MACE (Figure 1D).
DISCUSSION
Baseline Data
We found a lower prevalence of previous CABG in our population (5.6%) of unselected ACS patients, in contrast with rates of 20% previously reported in observational studies,6,7 but similar to the data most recently published by the Valiant trial (7%).17 In addition, it is worth noting that the prevalence of previous percutaneous revascularization was higher (9.6%, data not shown) for the overall population, probably reflecting an overall change in the treatment of coronary artery disease
As in other published series, our patients with a history of CABG were older, more often male, and more likely to have diabetes and a history of PCI and MI. This could be explained by adherence to guidelines in our series regarding surgical revascularization, and by the fact that this patient subgroup has a longer history of atherosclerotic disease.
The time between CABG and the ACS was similar to that previously reported in the literature,18 and varied between a median of 4 to 12.5 years.
Patients with a history of CABG were admitted more often for a non-ST-elevation ACS and also had lower peak values of cardiac biomarkers for necrosis. This has also been previously reported and is probably related to the importance of the dual circulation (a protection mechanism for patients with ACS and a history of CABG). The protection conferred by the bypass vessels to the main coronary arteries and the more common occlusion of secondary branches as the cause of the ACS, as reported by Crean et al19 in 1985, could explain this finding.
Prognosis
In our population, although a history of CABG was associated in absolute terms with a poorer outcome in terms of in-hospital and 1-year mortality, the results were not statistically different. A higher number of patients and a longer follow-up could probably have enhanced the difference and, therefore, allowed us to conclude (as other authors have10,17,20-22) that patients with a history of CABG have a poorer outcome when they have an ACS (although a history of CABG was not an independent predictor of outcome, as assessed by the GRACE and the Valiant cohort studies). Nevertheless, we considered the following hypothesis to explain this more favorable outcome among our population with ACS and a history of CABG, when compared to previous data in the literature:
- The high prevalence of previous treatment with aspirin (78.9%), statins, beta blockers, and ACE inhibitors (50% in all 3 cases) before the current ACS. This rate was higher than the one published by the TIMI group,10 in which only 60% of patients were receiving aspirin and only 20% of patients were on ACE inhibitors, but comparable to a subanalysis of the GRACE registry.23 This finding reinforces the importance of the long-term protective effect achieved with these 4 drug classes.
- Adherence to medical treatment guidelines were quite high in our patient subgroup, both in-hospital and at discharge, with higher levels compared to the European Heart Surveys in 2004.24
- Another important aspect of our analysis was aggressive antiplatelet therapy (53.4%), especially with GP IIb/IIIa inhibitors. Only 19% of the Valiant trial population with a history of CABG were offered GP IIb/IIIa.17 We believe these drugs are a crucial treatment for dealing with unstable coronary lesions, regardless of the culprit lesion, as previously reported by the subgroup analysis of the PRISM-PLUS and PURSUIT trial.21
- Similar rate of primary PCI between groups. In the Valiant trial, only 14% of patients with a history of CABG underwent PCI.
Revascularization
Almost 50% of patients with a history of CABG received an invasive strategy in the hospital, and most interventions were performed in native vessels with DES. In particular, there were multiple anatomic possibilities for the culprit lesion in these patients, and the ECG may not be that helpful in defining the location of the ischemia. Therefore, the decisions regarding revascularization in CABG were difficult, as the harm from a percutaneous intervention (especially because we were dealing with a complex lesion) could upset the delicate balance of native versus graft circulation. According to the data from a GRACE substudy, published by Gurfinkel et al,23 on the benefits of a conservative versus invasive strategy for the patient subgroup with a history of CABG, there was no benefit based on the 6-month adjusted data, and a greater proportion of patients in the invasive group required unscheduled and therapeutic invasive procedures, which meant that revascularization leads to further revascularization (although angiographic data on the coronary lesions were not reported).
Moreover, data on percutaneous revascularization of saphenous venous grafts are still debated. The literature reports on randomized studies comparing stents versus balloon angioplasty for saphenous vein stenosis, which showed no statistical difference in restenosis during a 6-month period, but with an improved clinical outcome for elective stenting.25 A recent Spanish study on 237 saphenous graft stenoses treated by DES or bare-metal stent (BMS) concluded a similar 30-month prognosis for both groups.26 Another study analyzed outcomes after PCI for patients with a history of CABG versus no history of CABG found that post-CABG patients had higher levels of event at 6 months, including death and MI, an outcome that was poorer for percutaneous transluminal coronary angioplasty (PTCA) in grafts versus native lesions. This poorer outcome could not be explained only by the patients' baseline characteristics, but also because graft PCI was difficult and the lesions were complex and contained more luminal thrombi.27 In a recent retrospective study by Lei Ge et al,28 the benefits of PCI with a DES vs. BMS were established for venous graft stenoses (6-month MACE-free survival of 88.5% for DES vs 71.9% for BMS), and the use of BMS was an independent predictor of MACE. In this study, the authors reported a low rate of total occlusions, only 4.3% for DES and 3.3% for BMS, and distal protection devices were used in a third of patients. In contrast, data from a randomized trial (with angiographic control) that compared sirolimus DES with BMS for similar lesions found significant benefit for DES regarding in-stent loss and target vessel revascularization, but with no major difference for the clinical endpoints.29 In this trial, patients with left ventricular dysfunction, renal impairment, and totally occluded graft lesions were excluded. Despite these benefits, a recent retrospective study for DES in saphenous grafts used in actual clinical practice reported a high rate of adverse events at 1 year, and the authors identified 2 independent predictors of adverse events (death and infarction at 1 year): luminal thrombus and the length of the stented segment.30 A meta-analysis comparing DES vs BMS for saphenous grafts showed a lower incidence of major adverse cardiac events in the DES group over a 6- to 12-month period, mainly due to a reduction in target vessel revascularization.31 Nevertheless, there is a paucity of data on PCI in grafts, whether in a stable or unstable condition, and few randomized trials of insufficient power and short-term follow-up.
In our previous subgroup of patients with ACS and a history of CABG, we chose full adherence to medical treatment guidelines, as well as percutaneous revascularization whenever technically feasible, preferably in native vessels with a DES, based on previously reported data. Our venous grafts presented a very high proportion of total occlusion ("real world") that limited PCI. In addition, no patients underwent a second surgical revascularization.
Finally, a history of CABG was an independent predictor for a future readmission for unstable angina. This fact could have been related to more severe ischemia that probably was not amenable to revascularization, but curiously was not synonymous with a poorer medium-term prognosis. We believe, therefore, that this cohort of ACS patients represented a good example of the long-term benefit of ischemic preconditioning and the collateral circulation which is the natural course of long-term atherosclerotic disease process.
Limitations
Our study included a small number of patients with ACS and a history of CABG ACS. We compared 2 groups that had important baseline differences, and the statistical results could be misleading because the study was underpowered (power of 35.5% for inhospital mortality, 3.6% for 1-year mortality, 12.3% for 1-year MACE, and 50% for readmission due to UA). However, we had no data on medication during follow-up, and the observational period was limited to 12 months. Lastly, 6.3% of patients were lost to follow-up.
CONCLUSIONS
In our study population, a history of CABG was an independent predictor of hospital readmission for UA 1 year after the ACS, although it did not significantly influence survival or other ischemic endpoints in the short or medium term. These data stress the need for aggressive medical treatment in this high-risk ACS population.
ABBREVIATIONS
ACS: acute coronary syndrome
BMS: bare-metal stent
CABG: coronary artery bypass graft
DES: drug-eluting stent
MI: myocardial infarction
NSTEMI: non-ST-elevation myocardial infarction
STEMI: ST-elevation myocardial infarction
UA: unstable angina
See editorial on pageS 516-7
Correspondence: Dr. R. Paiva Cardoso Teixeira.
Serviço de Cardiología, Hospitais da Universidade de Coimbra, Praceta Prof. Mota Pinto, 3000 - 050 Coimbra, Portugal
E-mail: rogeriopteixeira@gmail.com
Received July 25, 2009.
Accepted for publication November 30, 2009.