To the Editor:
Alport syndrome is a heterogeneous genetic disease caused by mutation in the genes that codify the type IV collagen of the basement membrane, which produces an overexpression of proteins in the extracellular matrices of the basement membrane, leading to glomerulosclerosis, which principally affects the kidneys, eyes, and inner ear.
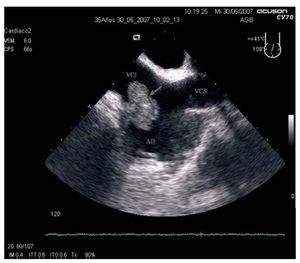
We present the case of a 35-year-old male with hypertension and chronic kidney failure caused by Alport syndrome, on haemodialysis since having rejected kidney transplants on 2 occasions, and fitted with a permanent central venous catheter as other vascular access was not possible. He was admitted with a profile of septic shock with suspected endocarditis or an infection site at the central catheter. Transaesophageal and transthoracic echocardiograms were taken, in which there were no signs of endocarditis, but which showed a mass of 33´23 mm along the right atrial wall and the Eustachian valve at the height of the inferior vena cava; the mass was nearly immobile, pedicled, and not calcified, and did not figure on the echocardiogram as a cardiac thrombus (Figure 1). Heparin, intravenous antibiotherapy and fluids were administered; with this treatment, the patient was stable and without fever in 48 hours. In the 20 days that followed, he underwent echocardiograph tests without any observable changes in the mass. For this reason, we opted for a surgical intervention and extracted a fragment approximately 25 mm in diameter with an excrescent area of 20´15´14 mm with a whitish colour, from the wall of the right atrium at the union of the inferior vena cava. Under the microscope, we observed an amorphous eosinophilic material with extensive calcifications, with fibrous tissue containing small vessels from hypertrophic walls and stromal micro-haemorrhage in the base (Figure 2). The anatomopathologic diagnosis was of an intraatrial tumor consistent with calcified amorphous tumor (CAT). The postoperative period was without complications. A postoperative echocardiograph test was performed, which detected no remains of the extirpated tumourous mass. Two months later, the patient remained asymptomatic.
Figure 1. Image from the transesophageal echocardiogram, caval view, in which the intracardiac mass (arrow) can be seen attached to the right atrial wall, near the union of the inferior vena cava (IVC).
Figure 2. Anatomopathological view of the cardiac wall with densely collagenised fibrous tumour growth (white arrow), with a good blood supply and with densely eosinophilic homogeneous central masses and focal dystrophic calcifications (black arrow).
CAT, described for the first time in 1997,2 is an infrequent cause of non-neoplastic intracardiac mass. Histologically, it is characterised by nodular calcium deposits over a matrix of fibrin and/or amorphous fibrin-like material, hyalinisation, inflammatory cells, and degenerated haematologic elements. It has been linked to organised thrombi, but its precise aetiology is unknown.3 Clinical tests usually show them to be benign, although they may cause obstruction or embolism,4 and cases can evolve fatally.5 The presence of cardiac CAT in haemodialysis patients has been described.6 During differential diagnosis, cardiac neoplasias, especially myxomas, and fibromas are considered, particularly if they are calcified,7 and so are conditions involving infection or thrombosis; in our case, there was no histologic data involving any of these possibilities, nor of other benign neoplasias. Calcified cardiac tuberculomas are infrequent and there is usually a history of tuberculosis,5 which was not the case for our patient. Cardiac tophaceous pseudogout and tumoral calcinosis are 2 rare entities associated with chronic kidney disease. However, tumoral calcinosis commonly appears as a dense calcium extension on the mitral ring and/or the aortic arch, with systemic calcification in non-skeletal tissue,5,7 which was not observed during the study of our patient.