To the Editor:
A 48-year-old male with a history of subarachnoid hemorrhage in 1992, cerebellar infarction due to vertebral artery occlusion with subsequent epilepsy was admitted to area hospital for cardiac catheterization after preceding 3 episodes of recurrent chest pain with signs of non-ST elevation myocardial infarction (NSTEMI). Transthoracic echocardiogram showed thin, akinetic apical segments of the left ventricle with scar extending into the middle segment of interventricular septum. Left ventricular ejection fraction was 35% and scarred apex contained 2 mural thrombi up to 16 mm diameter with spontaneous echocontrast filling the adjacent area. A peculiar color flow pattern was noted within normal myocardium (accelerated >100cm/s, predominantly diastolic flow) suggesting excessive collateralization with reversed apical coronary flow (Figure 1). Proximal right coronary artery was dilated (10 mm) whereas the left main coronary artery could not be visualized in typical situs. Coronary arteriography (Figure 2) demonstrated a unique coexistence of 2 rare coronary anomalies. A massively dilated left anterior descending artery (LAD) originated from the main pulmonary artery. LAD flow was reversed due to collateral supply through large (6 mm diameter) vessels conducting blood from the right and circumflex artery. The right coronary artery was extremely dilated (10 mm). The circumflex branch of the left coronary artery (CX) had an abnormal origin from the right coronary sinus and the further course was posterior (behind the aortic root). There were no signs of CX compression or excessive angulation resulting in luminal narrowing.
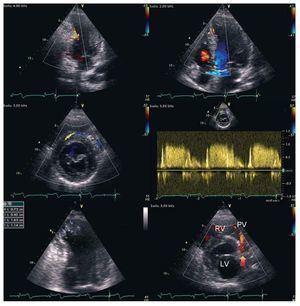
Figure 1. Transthoracic echocardiography. Top panels: color-coded flow within left ventricular wall indicates accelerated flow in dilated right coronary artery (left) and septal collaterals (right). Middle panel: parasternal short axis view demonstrating a large collateral vessel with flow directed frontally from the right coronary artery towards the left anterior descending coronary artery with peak diastolic velocity above 100cm/s according to pulsed wave spectral Doppler. Bottom left: thrombi seen in the infarcted apex of the left ventricle (arrow). Bottom right: retrograde diastolic flow (coded in red, arrow) from the left anterior descending artery towards the pulmonary valve (PV). LV indicates left ventricle; RV, right ventricle.
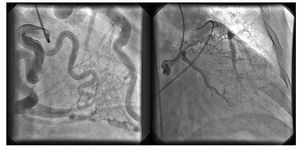
Figure. 2. Coronary angiogram in RAO 30o view. Left: contrast injection into the dilated right coronary artery (RCA) demonstrates retrograde filling of the left anterior descending coronary artery via extensive septal collaterals. Right: contrast injection in the circumflex branch with retrograde filling of the left anterior descending coronary artery through marginal-diagonal collaterals. Retrograde flow of contrast into the main pulmonary artery (with Amplatz catheter in the lumen) is evident.
Due to recurrent NSTEMI episodes and angiographically defined anomaly, the patient was referred for viability assessment. MRI delayed enhancement study demonstrated a transmural scar in LAD territory. The patient has been remaining asymptomatic on medical treatment since then.
The finding of extremely dilated coronary arteries and collaterals is typical for Bland-Garland-White syndrome or ALCAPA (anomalous left coronary artery from pulmonary artery), entity extremely rare in adults (estimated 1:300 000 live births).1 Coexistence of another coronary anomaly is even more unusual - there are several reports on isolated LAD origin from pulmonary artery with circumflex branch originating in the left sinus of Valsalva2,3 but to our knowledge only 2 cases of ALCAPA with CX originating from the right sinus of Valsalva4,5 have been described. However, none of them reported unique Doppler flow features demonstrated in transthoracic echocardiogram in our patient.
As the area supplied by pulmonary artery is profoundly ischemic and usually becomes infarcted in childhood,6 concomitant CX anomaly protects a subregion of left coronary territory supplied by the circumflex branch. In our patient anomalous circumflex branch has thus became the second source of oxygenated blood inflow in the left coronary region. Left-to-right shunt is another aspect of the problem and Qp/Qs ratio in our patient was below 1.5 (transthoracic echocardiographic calculation); therefore, considering the absence of viability in LAD territory, no intervention was undertaken. Otherwise, possible treatment options might include surgical reimplantation of LAD into the aortic sinus or embolization of LAD (to terminate shunt in case of non-viable LAD territory).
Multimedia material additional (video) available at: www.revespcardiol.org