To the Editor:
We present the case of a female patient aged 65 who underwent an aortic valve replacement in 2002 with a different surgical team from our own and received a size-16 mechanical prosthesis. Ultrasound observation revealed high transprosthetic gradients (mean, 39 mm Hg; max, 82 mm Hg) and a functional area of 1 cm2, with pulmonary hypertension (61 mm Hg). In previous months, she had required admission twice due to congestive heart failure.
We considered replacing the prosthetic aortic valve. Given the small diameter of the native valvular ring and the high risk of reoperation (logistic EuroSCORE, 23.38), we opted for implanting an apicoaortic conduit.
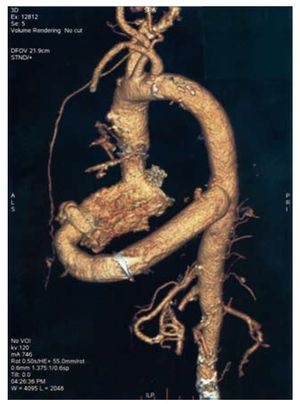
After cannulating the left femoral artery to begin complete cardiopulmonary bypass, we performed a left lateral thoracotomy through the fifth intercostal space, opening the pericardium. Next, we prepared the descending thoracic aorta. After switching on bypass, a lateral aortic clamp was placed to create a terminolateral anastomosis with a 21mm valved tube by St Jude Medical®. Next, a 2 cm lateral opening was made in the left ventricular apex with a 20 mm Medtronic® circular blade and the 20 mm Medtronic® apicoaortic connector was introduced and attached to the ventricular apex with eight prolene sutures reinforced with Teflon patches. Following that step, the apicoaortic connector was sutured end-to-end to the valved tube that had been anastomosed to the descending thoracic aorta (Figure).
Figure 1. Computerised tomography of the apicoaortic conduit.
Six weeks after the surgery, the patient's evolution was good and her exercise tolerance had increased.
Prosthesis-patient mismatch (PPM) was first described by Rahimtoola1 in 1978 as a situation in which the functional valvular area following aortic valve replacement is less than that of a normal human valve. Studies that have analysed PPM's effect on mortality2,3 and its haemodynamic repercussions have shown variable results.4 In our patient, the relative functional area for the aortic prosthesis was 0.55 cm2/m2, which is considered severe PPM.5
Since the first case of implanting an apicoaortic conduit was published in 1975,6 the technique has been successfully used for congenital aortic valve and LV outflow tract anomalies7 as well as acquired aortic valve conditions. These include previous surgical procedures with permeable grafts, complex left ventricular outflow tract lesions and porcelain aorta.8-11
The apicoaortic conduit offers advantages with respect to conventional valve repair with aortic ring enlargement. Firstly, it circumvents the need for a second sternotomy. In addition, the conduit is placed while the heart is beating, which avoids myocardial ischaemia in a high-risk patient. However, the procedure may be technically difficult, particularly in patients with friable tissues. Embolic complications have also been described in conduits containing biological prostheses.9
To our understanding, this is the first published case of this technique being successfully used to treat a case of PPM for which a traditional valve replacement would have entailed a high level of risk and a low probability of good results.