We thank Blasco-Lucas et al.1 for their interest in our article. We agree with them that the first firm evidence of the high incidence of structural valve deterioration (SVD) in the Mitroflow prosthesis came from the article by the French group.2 Three years later, we added a larger series, with a longer follow-up and an analysis that estimated the true cumulative incidence of the event (SVD) in the presence of a high percentage of competing events (essentially, death).
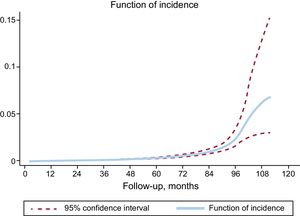
Analyzing the data again, we can graphically represent (Figure) the rate (incidence) at which SVD occurred and confirm that it increases from the fifth year and, in particular, from the seventh.
Several characteristics differentiate this prosthesis from others, and while the lack of anticalcification treatment is one of them, it is not the only one. Its architecture, with some pericardial leaflets mounted externally to the stent, improved the hemodynamic profile of the existing valves at the start of the 1980s. However, this could cause pressure to be absorbed principally by the leaflets and not by the device skeleton. If this were important, the absence or presence of anticalcification treatment would be irrelevant.
More than 30 years were needed to obtain firm evidence of the increased incidence of SVD in this prosthesis. Before that study,2 several studies had been published that “demonstrated” exactly the opposite. Publishing negative results on medical devices appears to be somewhat more difficult than publishing positive results. A similar problem worried the epidemiologist Rosenthal when he described “the file drawer problem”: the fact that the medical literature is full of the 5% of studies that have a type I error, while the authors’ file drawers are full of the other 95%.3